- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
OCR A Level Biology:复习笔记2.4.6 Enzyme Activity: Substrate Concentration
Enzyme Activity: Substrate Concentration
- The greater the substrate concentration, the higher the rate of reaction:
- As the number of substrate molecules increases, the likelihood of enzyme-substrate complex formation increases
- If the enzyme concentration remains fixed but the amount of substrate is increased past a certain point, however, all available active sites eventually become saturated and any further increase in substrate concentration will not increase the reaction rate
- When the active sites of the enzymes are all full, any substrate molecules that are added have nowhere to bind in order to form an enzyme-substrate complex
- For this reason, in the graph below there is a linear increase in reaction rate as substrate is added, which then plateaus when all active sites become occupied
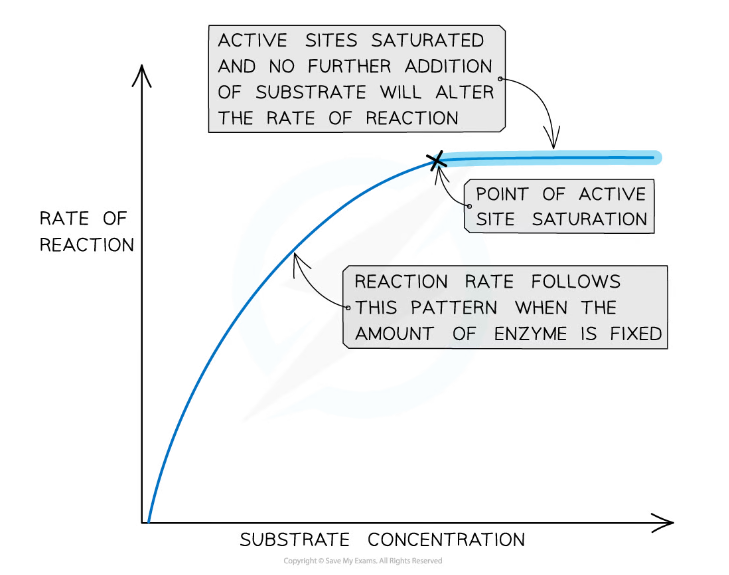
The effect of substrate concentration on the rate of an enzyme-catalysed reaction
Exam Tip
If substrate concentration is continually increased but enzyme concentration is kept constant, there eventually comes a point where every enzyme active site is working continuously. At this point, the substrate molecules are effectively ‘queuing up’ for an active site to become available.At this stage, the enzyme is working at its maximum possible rate, known as Vmax (V stands for velocity).
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





