- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
OCR A Level Biology:复习笔记2.2.14 Inorganic Ions
Inorganic Ions
- An ion is an atom (or sometimes a group of atoms) that has an electrical charge
- An ion that has a +ve charge is known as a cation
- An ion that has a -ve charge is known as an anion
- An inorganic ion is an ion that does not contain carbon
- Inorganic ions play an important role in many essential cellular processes
- Inorganic ions occur in solution in the cytoplasm and body fluids of organisms
- Some occur in high concentrations and others in very low concentrations
- The concentration of certain ions can fluctuate and can be used in cell signalling and neuronal transmission
- Each type of inorganic ion has a specific role, depending on its properties
- Some inorganic ions act as cofactors
- Cofactors are non-protein chemical compounds that are required for a protein to function
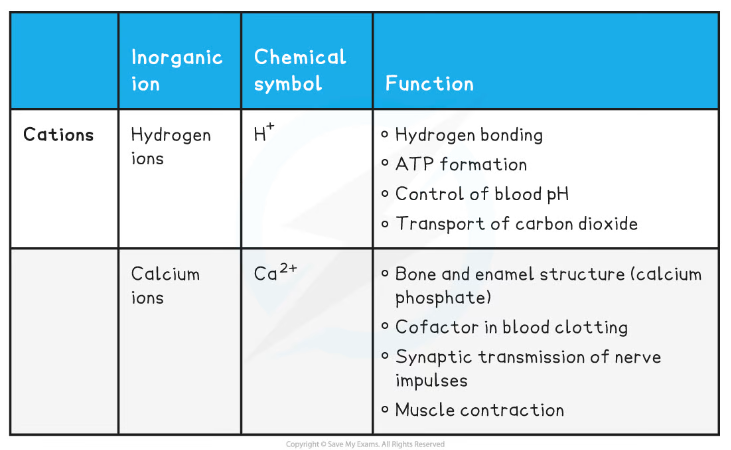
- You should know the following inorganic ions, as well as their properties and roles in the body:
- Hydrogen ions (H+)
- Calcium ions (Ca2+)
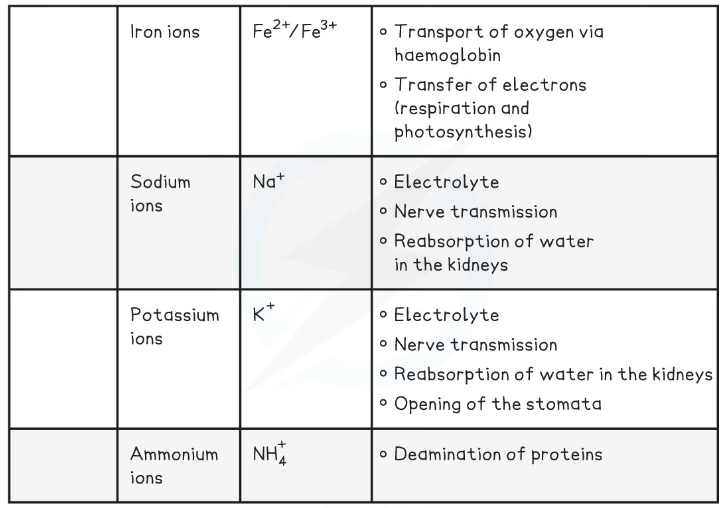
- Iron ions (Fe2+/Fe3+)
- Sodium ions (Na+)
- Potassium ions (K+)
- Ammonium ions (NH4+)
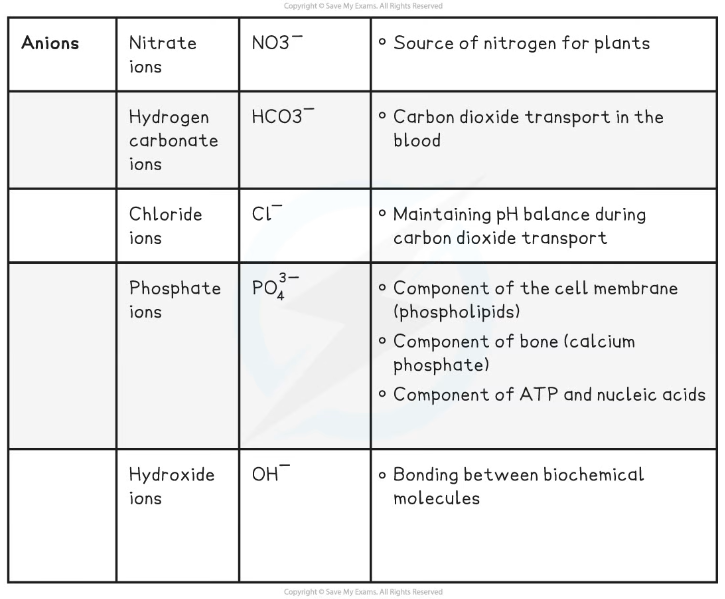
- Nitrate ions (NO3-)
- Hydrogen carbonate ions (HCO3-)
- Chloride ions (Cl-)
- Phosphate ions (PO43-)
- Hydroxide ions (OH-)
Hydrogen ions
- Hydrogen ions are protons
- The concentration of H+ in a solution determines the pH
- There is an inverse relationship between the pH value and the hydrogen ion concentration
- The more H+ ions present, the lower the pH (the more acidic the solution)
- The fewer H+ ions present, the higher the pH (the more alkaline the solution)
- The concentration of H+ is therefore very important for enzyme-controlled reactions, which are all affected by pH
- The fluids in the body normally have a pH value of approximately 7.4
- The maintenance of this normal pH is essential for many of the metabolic processes that take place within cells
- Changes in pH can affect enzyme structure
- For example, abnormal levels of hydrogen ions can interact with the side-chains of amino acids and change the secondary and tertiary structures of the proteins that make up enzymes
- This can cause denaturation of enzymes
Calcium ions
- Ca2+ is essential in the movement of organisms:
- In synapses, calcium ions regulate the transmission of impulses from neurone to neurone
- Ca2+ also stimulates muscle contraction
- When an impulse reaches a muscle fibre, Ca2+ is released from the sarcoplasmic reticulum
- This Ca2+ binds to troponin, removing the tropomyosin from myosin-binding sites on actin
- This allows actin-myosin cross-bridges to form when the muscle fibre contracts
- Ca2+ can also help to regulate protein channels, which affects the permeability of cell membranes
- Many enzymes are activated by Ca2+, making these ions key regulators in many biological reactions
- The presence of Ca2+ is also necessary for the formation of blood clots (it is known as a clotting factor or a cofactor)
Iron ions
- There are actually two versions of iron ions (known as oxidation states)
- Iron (II) ions, also known as ferrous ions (Fe2+)
- Iron (III) ions, also known as ferric ions (Fe3+)
- Iron ions are essential as they can bind oxygen
- Haemoglobin is the large protein in red blood cells that is responsible for transporting oxygen around the body
- Haemoglobin is made up of four polypeptide chains that each contain one Fe2+
- This Fe2+ is a key component in haemoglobin as it binds to oxygen
- Myoglobin in muscles functions in a similar way (it is an oxygen-binding protein) but is only made up of one polypeptide chain (containing one Fe2+)
- Iron ions are also essential as they are involved in the transfer of electrons during respiration and photosynthesis, so they are key to the biological generation of energy
- Iron ions are an essential component of cytochromes (that are themselves a component of electron transport chains)
- Cytochrome c contains an iron ion that is essential to its function
- During the electron transport process, this iron ion switches between the Fe3+ and Fe2+ oxidation states, which allows for electrons to be accepted and donated
Sodium ions
- Na+ is required for the transport of glucose and amino acids across cell-surface membranes (e.g. in the small intestine)
- Glucose and amino acid molecules can only enter cells (through carrier proteins) alongside Na+
- This process is known as co-transport
- First, Na+ is actively transported out of the epithelial cells that line the villi
- The Na+ concentration inside the epithelial cells is now lower than the Na+ concentration in the lumen of the small intestine
- Na+ now re-enters the cells (moving down the concentration gradient) through co-transport proteins on the surface membrane of the epithelial cells, allowing glucose and amino acids to enter at the same time
- Na+ is also required for the transmission of nerve impulses
Potassium ions
- K+ is essential for nerve transmission
- Potassium ion voltage-gated channel proteins in a section of axon membrane open, allowing the diffusion of potassium ions out of the axon, down their concentration gradient. This returns the potential difference across the axon membrane back to normal (about -70mV) – a process known as repolarisation
- K+ allows for the reabsorption of water in the kidneys
- Potassium ions also play an important role in guard cells and the opening of the stomata
Ammonium ions
- NH4+ is the intermediate ion that forms during the deamination of proteins in the liver and kidneys
Nitrate ions
- NO3- ions are present in the soil and are taken up by plants
- NO3- provides an essential source of nitrogen for protein synthesis
- Proteins are required for growth and repair in plants
Hydrogen carbonate ions
- HCO3- ions work alongside hydrogen ions in the transport of carbon dioxide in the blood
Chloride ions
- Cl- ions are also involved in the transport of carbon dioxide in the blood
- These ions move in and out of red blood cells and help to maintain pH balance
Phosphate ions
- PO43- attaches to other molecules to form phosphate groups, which are an essential component of DNA, RNA and ATP
- In DNA and RNA, the phosphate groups allow individual nucleotides to join up (to form polynucleotides)
- In ATP, the bonds between phosphate groups store energy
- These phosphate groups can be easily attached or detached
- When the bonds between phosphate groups are broken, they release a large amount of energy, which can be used for cellular processes
- Phosphates are also found in phospholipids, which are key components of the phospholipid bilayer of cell membranes
Hydroxide ions
- OH- ions play a vital role in bonding between biochemical molecules
- One oxygen atom is covalently bonded to one hydrogen atom
- The electronegative charge of the oxygen allows for the formation of hydrogen bonds
- Hydrogen bonds are important in many molecules
- DNA base pairing
- Secondary, tertiary and quaternary protein structure
- Water
Inorganic Ions Table
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





