- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
OCR A Level Biology:复习笔记2.2.13 Fibrous Proteins
Fibrous Proteins
- Fibrous proteins are long strands of polypeptide chains that have cross-linkages due to hydrogen bonds
- These proteins have little or no tertiary structure
- Due to a large number of hydrophobic R groups, fibrous proteins are insoluble in water
- Fibrous proteins have a limited number of amino acids with the sequence usually being highly repetitive
- The highly repetitive sequence creates very organised structures that are strong and this along with their insolubility property, makes fibrous proteins very suitable for structural roles
- Examples of fibrous proteins:
- Keratin makes up hair, nails, horns and feathers (it is a very tough fibrous protein)
- Elastin is found in connective tissue, tendons, skin and bone (it can stretch and then return to its original shape)
- Collagen is a connective tissue found in skin, tendons and ligaments
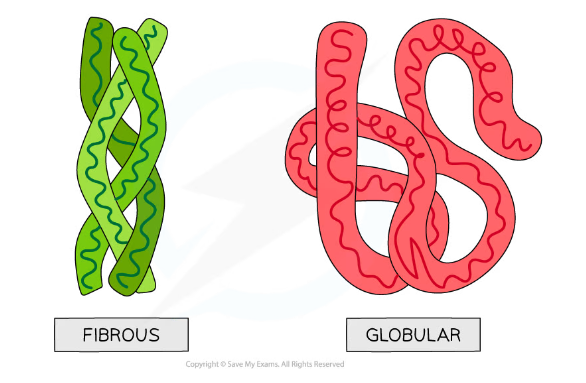
Globular and fibrous protein models illustrating the roughly spherical shape of globular proteins and the long, stranded shape of fibrous proteins
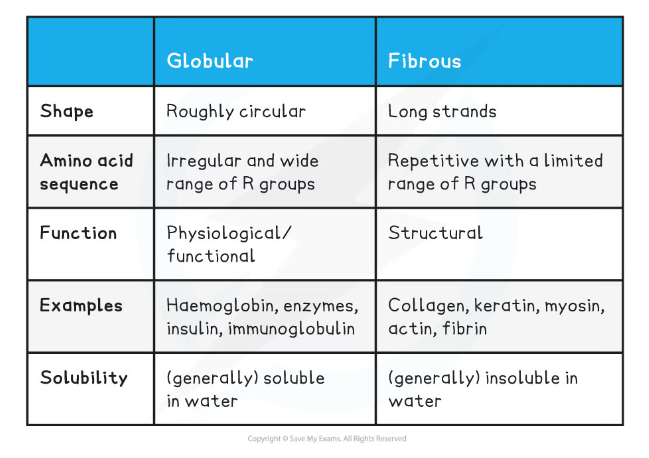
Comparison of Globular & Fibrous Tertiary Proteins Table
Collagen
- Collagen is the most common structural protein found in vertebrates
- It provides structural support
- In vertebrates it is the component of connective tissue which forms:
- Tendons
- Cartilage
- Ligaments
- Bones
- Teeth
- Skin
- Walls of blood vessels
- Cornea of the eye
- Collagen is an insoluble fibrous protein
Structure of collagen
- Collagen is formed from three polypeptide chains closely held together by hydrogen bonds to form a triple helix (known as tropocollagen)
- Each polypeptide chain is a helix shape (but not α-helix as the chain is not as tightly wound) and contains about 1000 amino acids with glycine, proline and hydroxyproline being the most common
- In the primary structure of collagen almost every third amino acid is glycine
- This is the smallest amino acid with a R group that contains a single hydrogen atom
- Glycine tends to be found on the inside of the polypeptide chains allowing the three chains to be arranged closely together forming a tight triple helix structure
- Along with hydrogen bonds forming between the three chains there are also covalent bonds present
- Covalent bonds also form cross-links between R groups of amino acids in interacting triple helices when they are arranged parallel to each other. The cross-links hold the collagen molecules together to form fibrils
- The collagen molecules are positioned in the fibrils so that there are staggered ends (this gives the striated effect seen in electron micrographs)
- When many fibrils are arranged together they form collagen fibres
- Collagen fibres are positioned so that they are lined up with the forces they are withstanding
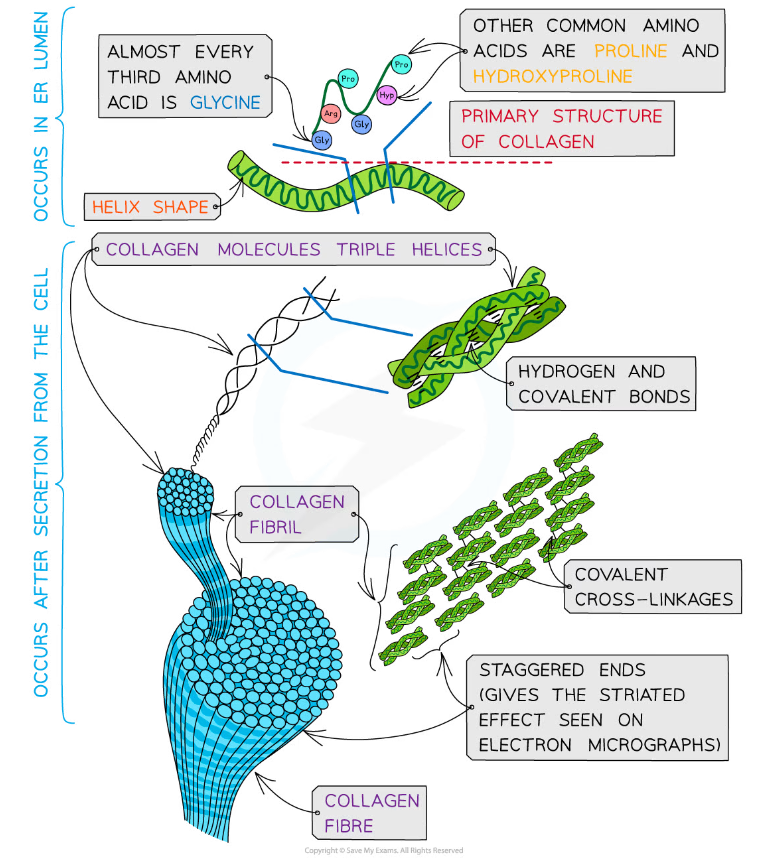
Collagen is a fibrous structural protein that is formed by triple helices collagen molecules arranging into collagen fibrils and finally into collagen fibres which have high tensile strength
Function of collagen
- Collagen is a flexible structural protein forming connective tissues
- The presence of the many hydrogen bonds within the triple helix structure of collagen results in great tensile strength. This enables collagen to be able to withstand large pulling forces without stretching or breaking
- The staggered ends of the collagen molecules within the fibrils provide strength
- Collagen is a stable protein due to the high proportion of proline and hydroxyproline amino acids present. These amino acids increase stability as their R groups repel each other
- The length of collagen molecules means they take too long to dissolve in water (making it insoluble in water)
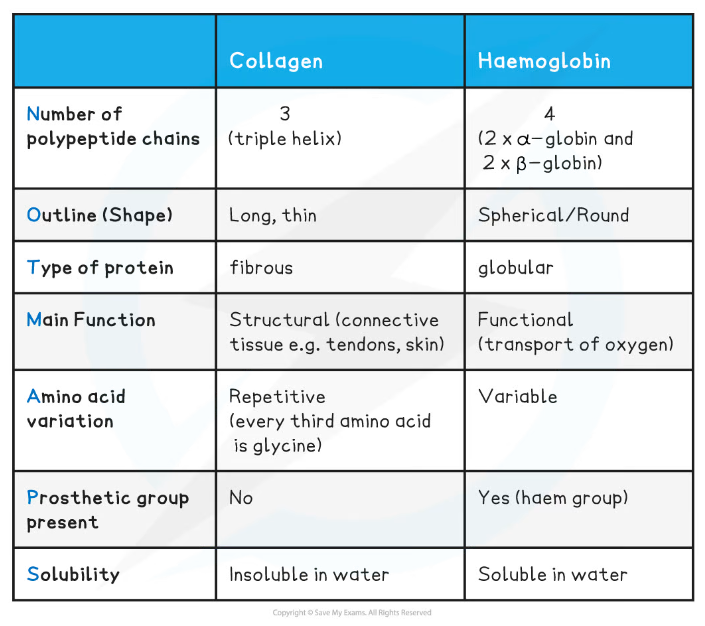
Comparison between Collagen & Haemoglobin Table
Exam Tip
To distinguish between proteins, learn SAFES (Shape, Amino acid sequence, Function, Examples and Solubility).You may also be asked to compare the structure and function of haemoglobin and collagen in the exam.
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





