- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
OCR A Level Biology:复习笔记2.1.2 Using a Microscope
Preparation of Microscope Slides
- Many biological structures are too small to be seen by the naked eye
- Optical microscopes are an invaluable tool for scientists as they allow for tissues, cells and organelles to be seen and studied
- For example, the movement of chromosomes during mitosis can be observed using a microscope
How optical microscopes work
- Light is directed through the thin layer of biological material that is supported on a glass slide
- This light is focused through several lenses so that an image is visible through the eyepiece
- The magnifying power of the microscope can be increased by rotating the higher power objective lens into place
Apparatus
- The key components of an optical microscope are:
- The eyepiece lens
- The objective lenses
- The stage
- The light source
- The coarse and fine focus
- Other tools used:
- Forceps
- Scissors
- Scalpel
- Coverslip
- Slides
- Pipette
- Staining solution
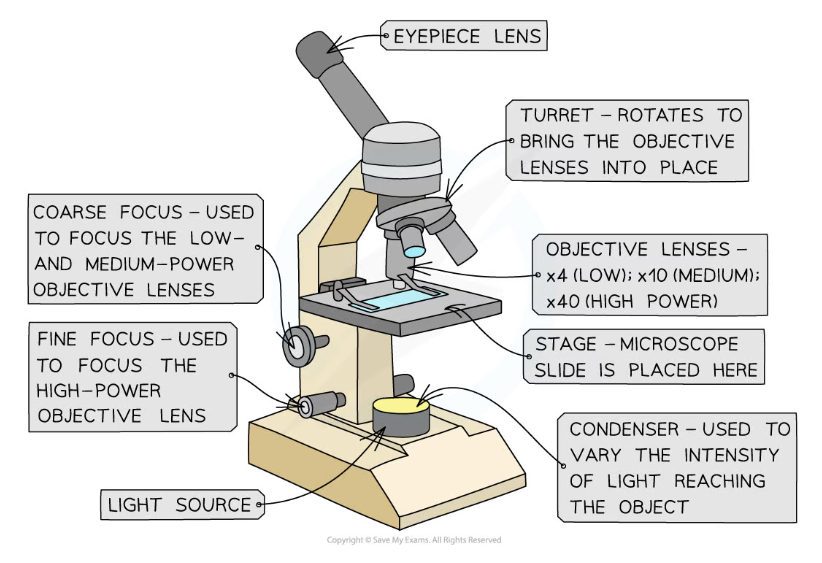
Image showing all the components of an optical microscope
Method
- Preparing a slide using a liquid specimen:
- Add a few drops of the sample to the slide using a pipette
- Cover the liquid/smear with a coverslip and gently press down to remove air bubbles
- Wear gloves to ensure there is no cross-contamination of foreign cells
- Methods of preparing a microscope slide using a solid specimen:
- Take care when using sharp objects and wear gloves to prevent the stain from dying your skin
- Use scissors to cut a small sample of the tissue
- Peel away or cut a very thin layer of cells from the tissue sample to be placed on the slide (using a scalpel or forceps)
- The tissue needs to be thin so that the light from the microscope can pass through
- Apply a stain
- Gently place a coverslip on top and press down to remove any air bubbles
Or
-
- Some tissue samples need to be treated with chemicals to kill/make the tissue rigid
- This involves fixing the specimen using formaldehyde (preservative), dehydrating it using a series of ethanol solutions, impregnating it in paraffin/resin for support then cutting thin slices from the specimen using a microtome
- The paraffin is removed from the slices/specimen, a stain is applied and the specimen is mounted using a resin and a coverslip is applied
Or
-
- Freeze the specimen in carbon dioxide or liquid nitrogen
- Cut the specimen into thin slices using a cryostat
- Place the specimen on the slide and add a stain
- Gently place a coverslip on top and press down to remove any air bubbles
- When using an optical microscope always start with the low power objective lens:
- It is easier to find what you are looking for in the field of view
- This helps to prevent damage to the lens or coverslip in case the stage has been raised too high
- Preventing the dehydration of tissue:
- The thin layers of material placed on slides can dry up rapidly
- Adding a drop of water to the specimen (beneath the coverslip) can prevent the cells from being damaged by dehydration
- Unclear or blurry images:
- Switch to the lower power objective lens and try using the coarse focus to get a clearer image
- Consider whether the specimen sample is thin enough for light to pass through to see the structures clearly
- There could be cross-contamination with foreign cells or bodies
- Using a graticule to take measurements of cells:
- A graticule is a small disc that has an engraved ruler
- It can be placed into the eyepiece of a microscope to act as a ruler in the field of view
- As a graticule has no fixed units it must be calibrated for the objective lens that is in use. This is done by using a scale engraved on a microscope slide (a stage micrometer)
- By using the two scales together the number of micrometers each graticule unit is worth can be worked out
- After this is known the graticule can be used as a ruler in the field of view
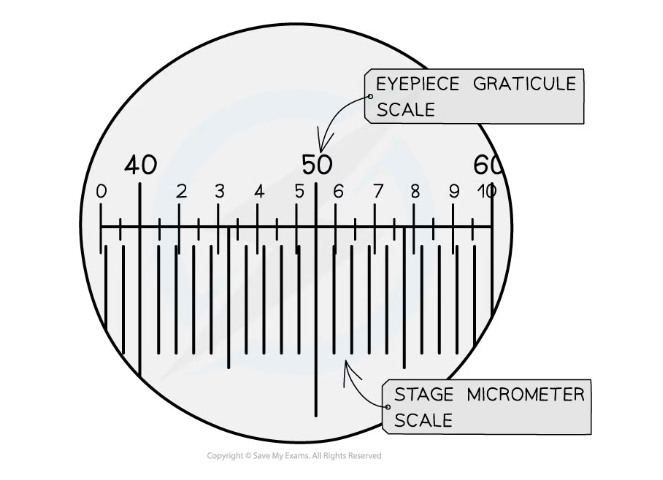
The stage micrometer scale is used to find out how many micrometers each graticule unit represents
Limitations
- The size of cells or structures of tissues may appear inconsistent in different specimen slides
- Cell structures are 3D and the different tissue samples will have been cut at different planes resulting in this inconsistencies when viewed on a 2D slide
- Optical microscopes do not have the same magnification power as other types of microscopes and so there are some structures that can not be seen
- The treatment of specimens when preparing slides could alter the structure of cells
Exam Tip
Remember the importance of calibration when using a graticule. If it is not calibrated then the measurements taken will be completely arbitrary!
Staining in Light Microscopy
- Many tissues that are used in microscopy are naturally transparent, they let both light and electrons pass through them
- This makes it very difficult to see any detail in the tissue when using a microscope
- Stains are often used to make the tissue coloured/visible
Staining for light microscopy
- Coloured dyes are used when staining specimens
- The dyes used absorb specific colours of light while reflecting others; this makes the structures within the specimen that have absorbed the dye visible
- Certain tissues absorb certain dyes, which dye they absorb depends on their chemical nature
- Specimens or sections are sometimes stained with multiple dyes to ensure the different tissues within the specimen show up - this is known as differential staining
- It is important to remember that most of the colours seen in photomicrographs (image taken using a light microscope) are not natural
- Chloroplasts don't need stains as they show up green, which is their natural colour
- Toluidine blue and phloroglucinol are common stains used
- Toluidine blue turns cells blue
- Phloroglucinol turns cells red/pink
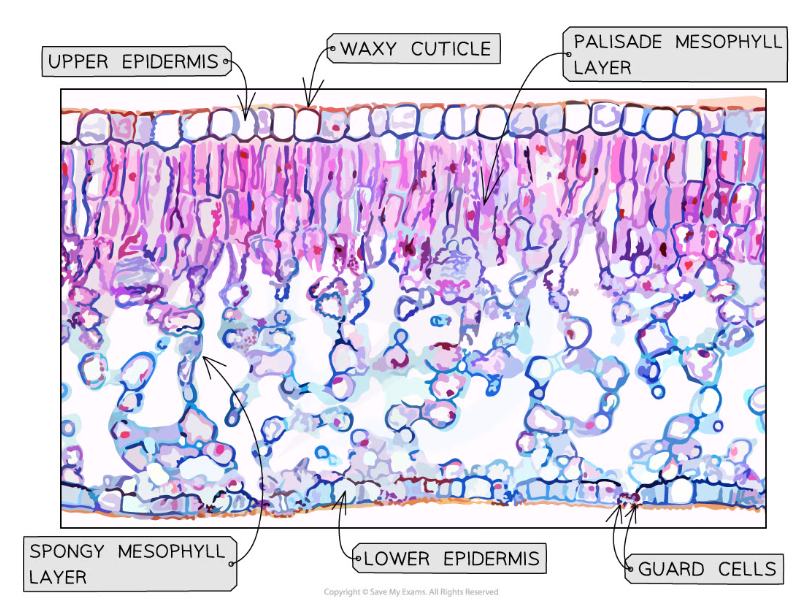
Toluidine blue and phloroglucinol have been used to stain this tissue specimen taken from a leaf
Staining for electron microscopy
- When using Transmission electron microscopes (TEMs) the specimen must be stained in order to absorb the electrons
- Unlike light, electrons have no colour
- The dyes used for staining cause the tissues to show up black or different shades of grey
- Heavy-metal compounds are commonly used as dyes because they absorb electrons well
- Osmium tetroxide and ruthenium tetroxide are examples
- Any of the colour present in electron micrographs is not natural and it is also not a result of the staining
- Colours are added to the image using an image-processing software
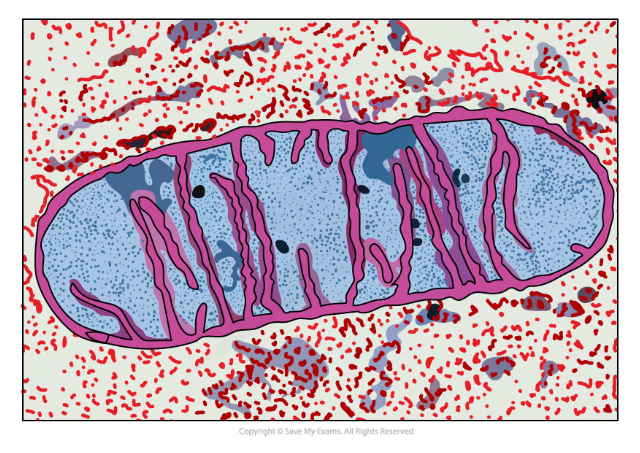
The internal structure of the mitochondrion can be seen using a TEM and staining

A spiracle found on the exoskeleton of an insect. No colours have been added to this image using image-processing software.
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





