- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A (SNAB) A Level Biology:复习笔记5.2.7 Practical: The Hill Reaction
Practical: The Hill Reaction
- The light-dependent reactions of photosynthesis take place in the thylakoid membrane and involve the release of high-energy electrons from chlorophyll molecules in photosystems
- These electrons are picked up by the coenzyme NADP along with hydrogen ions from photolysis, forming NADPH, or reduced NADP
- NADP is said to be the electron acceptor in this process
- The formation of NADPH in this way is known as the Hill reaction, named after scientist Robert Hill who discovered the process
- The rate of the Hill reaction can be studied using indicators such as DCPIP and methylene blue
- When present these indicators accept the electrons instead of NADP
- This causes the indicators to change colour
- Oxidised DCPIP (blue) → DCPIP accepts electrons → reduced DCPIP (colourless)
- Oxidised methylene blue (blue) → methylene blue accepts electrons → reduced methylene blue (colourless)
- Note that the colour of the reduced solution may appear green due to the presence of chlorophyll
- The rate at which the indicator changes colour from its oxidised (blue) state to its reduced (colourless) state can be used as a measure of the rate of the Hill reaction
- A faster colour change means a faster rate of reaction, and vice versa
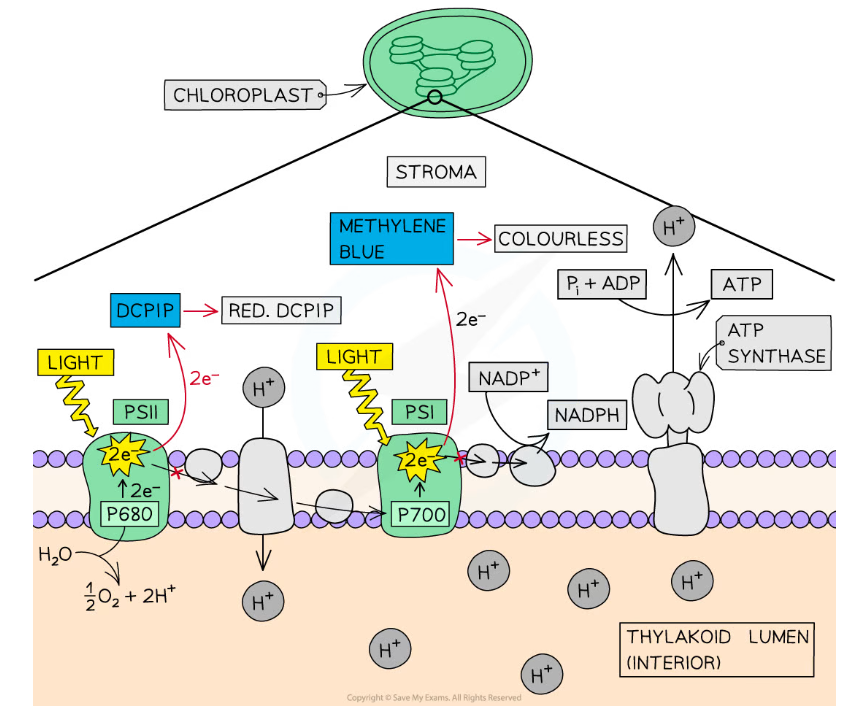
DCPIP and methylene blue are indicators that accept electrons, causing them to change from blue to colourless. The rate at which this colour change takes place indicates the rate at which the Hill reaction is taking place.
Investigating the rate of the Hill reaction
Apparatus
- Leaves e.g. spinach
- Pestle and mortar or food blender
- Isolation solution containing sucrose, potassium chloride and a pH 7 buffer
- Funnel
- Filter paper or cloth
- Beaker
- Centrifuge and centrifuge tubes
- Glass rods
- Ice-cold water bath
- Colorimeter and cuvettes
- Test tubes and rack
- Lamp
- DCPIP indicator
- Dropping pipette
Method
- Grind up the leaves with 20 cm3 isolation solution in a pestle and mortar or blend them in a food blender for 10 seconds
- This breaks apart the tissues of the leaf
- The isolation medium will prevent cell damage due to osmosis or extreme pH
- Filter the resulting liquid into a clean beaker using a funnel and some filter paper or cloth
- This removes large pieces of leaf tissue
- Transfer the filtered liquid into a centrifuge tube and centrifuge for 10 minutes
- This will result in a pellet of chloroplasts forming at the bottom of the tube
- Discard the liquid in the centrifuge tube and keep the pellet
- The liquid here is known as the supernatant
- Place 2 cm3 fresh isolation medium and the chloroplast pellet into a clean test tube, stirring with a glass rod to re-suspend the chloroplasts in the liquid; this is now the chloroplast extract
- Transfer the chloroplast extract to an ice-cold water bath
- The cold temperature of the water bath slows down the activity of the chloroplasts
- Place a test tube containing 0.5 cm3 chloroplast extract into a test tube rack set up at a set distance from a lamp
- A beaker of water can be placed in between the lamp and the rack here to prevent a temperature increase due to heat from the lamp
- Add 5 cm3 DCPIP solution to the chloroplast extract and mix together using a clean glass rod
- Use a pipette to immediately remove a sample of the DCPIP-chloroplast mixture and place the sample into a clean cuvette
- A cuvette is a small plastic container with clear, colourless sides designed for use in a colorimeter
- Place the cuvette into a colorimeter and take a reading for absorbance
- A colorimeter is a piece of equipment used to measure how much light passes through or is absorbed by a solution
- The amount of light that passes through is transmission while the light absorbed is absorbance
- Repeat steps 9-10 every minute for 10 minutes
- Repeat steps 7-11 at least twice more, ensuring that the distance from the lamp and the timings of the samples are kept the same
Results
- The absorbance reading of the colorimeter should decrease over the 10 minute test period as the DCPIP indicator is reduced by electrons in the chloroplasts
- The solution changes from blue to colourless as the DCPIP is reduced, reducing the amount of light that is absorbed by the solution
- A graph of absorbance against time can be plotted to show the rate of the Hill reaction
- Changing variables such as light intensity, light wavelength, or temperature of the chloroplast extract would allow the effect of different variables on the rate of the Hill reaction to be studied
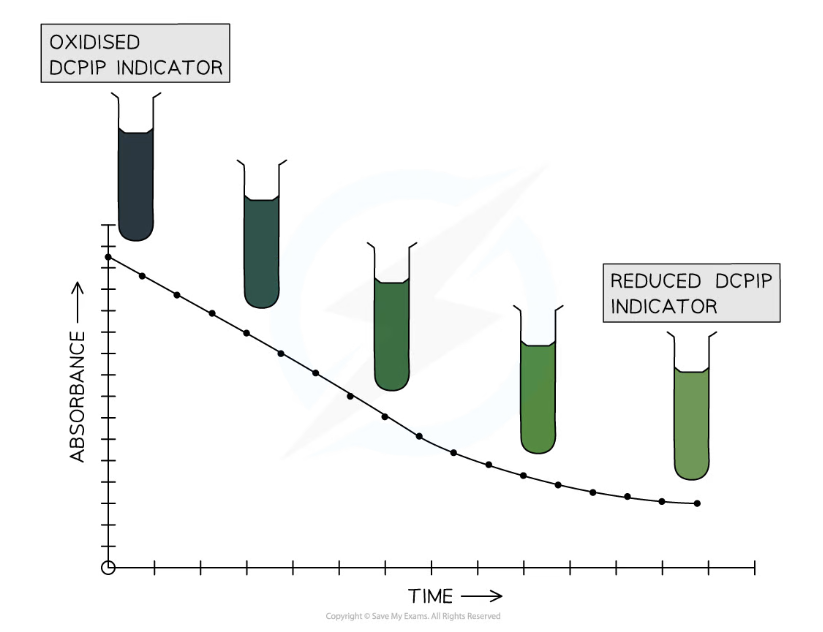
DCPIP indicator changes colour from blue to colourless as it is reduced. Note that the presence of chloroplasts in the solution causes it to appear green rather than colourless.
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





