- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel IGCSE Physics: Double Science 复习笔记:7.2.4 Contamination & Irradiation
Edexcel IGCSE Physics: Double Science 复习笔记:7.2.4 Contamination & Irradiation
Contamination & Irradiation
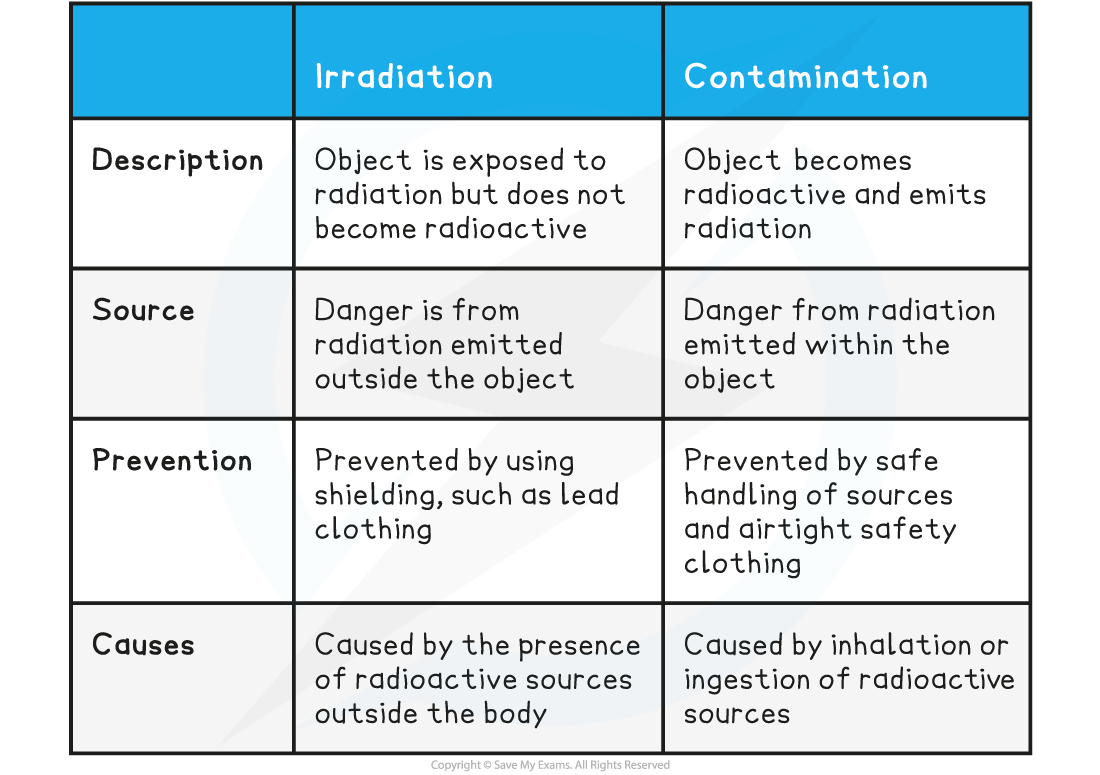
Contamination
- Contamination is defined as:
The unwanted presence of materials containing radioactive atoms on other materials
- A substance is only radioactive if it contains radioactive atoms that emit radiation
- Contamination occurs when a radioactive isotope gets onto a material where it should not be
- This is often due to a radiation leak
- As a result of this, the small amounts of the isotope in the contaminated areas will emit radiation and the material becomes radioactive
The Assassination of Alexander Litvinenko
- Contamination is almost always a mistake or an accident
- However, in 2006 a former Russian spy was poisoned by a radioactive isotope
- His name was Alexander Litvinenko and he was contaminated with the isotope polonium-210
- He died because of the poisoning

It is believed that the polonium-210 that poisoned Alexander Litvinenko was secretly put into a cup of tea he was drinking
Irradiation
- Irradiation is defined as:
The process of exposing a material to alpha, beta or gamma radiation
- Irradiating a material does not make that material radioactive
- However, it can kill living cells

This sign is the international symbol indicating the presence of a radioactive material
- Irradiation can be used as a method of sterilisation:
- Surgical equipment is irradiated before being used in order to kill any micro-organisms on it before surgery
- Food can be irradiated to kill any micro-organisms within it
- This makes the food last longer without going mouldy
Hazards of Irradiation and Contamination
- Although irradiation can cause harm, contamination has the potential to cause far more harm, due to the continuous exposure to radiation that it will produce
- Contamination is particularly dangerous if a radioactive source gets into the human body
- The internal organs will be irradiated as the source emits radiation as it moves through the body
Comparison of Irradiation and Contamination Table
Protecting against Irradiation and Contamination
- It is important to reduce the risk of exposure to radiation
- Radiation can mutate DNA in cells and cause cancer
- Shielding is used to absorb radiation
- Lead lined suits are used to reduce irradiation for people working with radioactive materials
- The lead absorbs most of the radiation that would otherwise hit the person
- To prevent contamination an airtight suit is used by people working in an area where there may have been a radiation leak
- This prevents radioactive atoms from getting inside the person
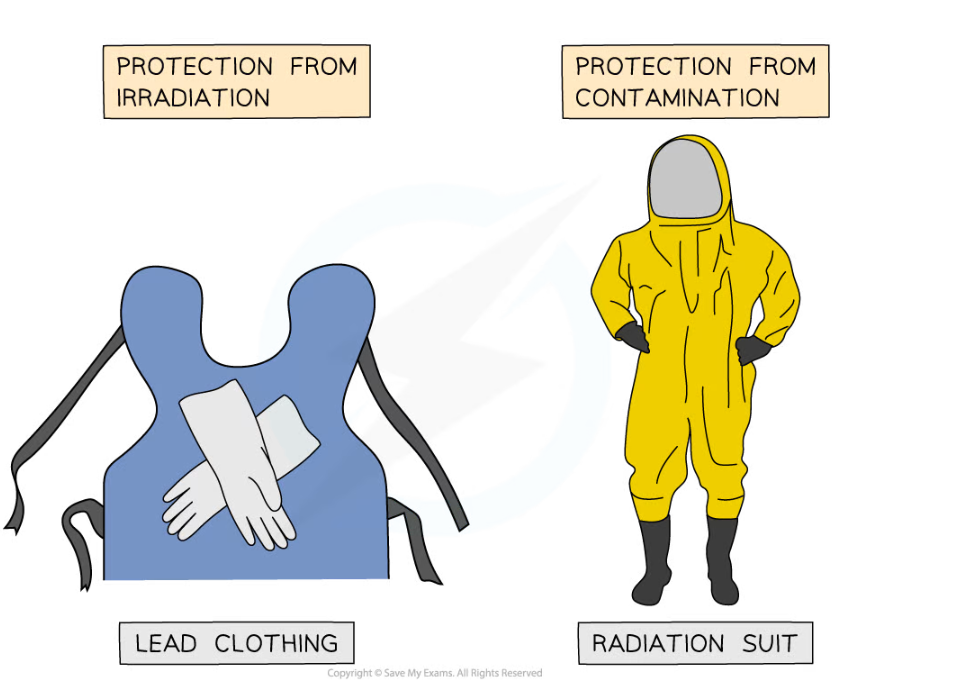
Lead shielding is used when a person is getting an x-ray, as well as for people who work with radiation. Contamination carries much greater risks than irradiation
Worked Example
Summarise the difference in the risk posed by radioactive sources with very short and very long half-lives with regards to:
(a) Irradiation.
(b) Contamination.
(a) Sources with short half-lives present a greater risk of irradiation
-
- A short half-life means a source has a high activity
- This means there is a high rate of radioactive emissions, compared to a source with a long half-life
(b) Sources with long half-lives present a greater risk of contamination
-
- Sources with long half-lives will remain radioactive for longer
- They need to be controlled for longer, to prevent them spreading
- Shielding and storage may be required
Exam Tip
Irradiation and contamination are very commonly confused.Remember that something is radioactive only if it contains radioactive atoms. This can only occur from contamination, not from irradiation!
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





