- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A Level Chemistry:复习笔记7.7.3 Proton NMR
Proton NMR - Introduction
- Nuclear Magnetic Resonance (NMR) spectroscopy is used for analysing organic compounds
- All samples are measured against a reference compound – Tetramethylsilane (TMS)
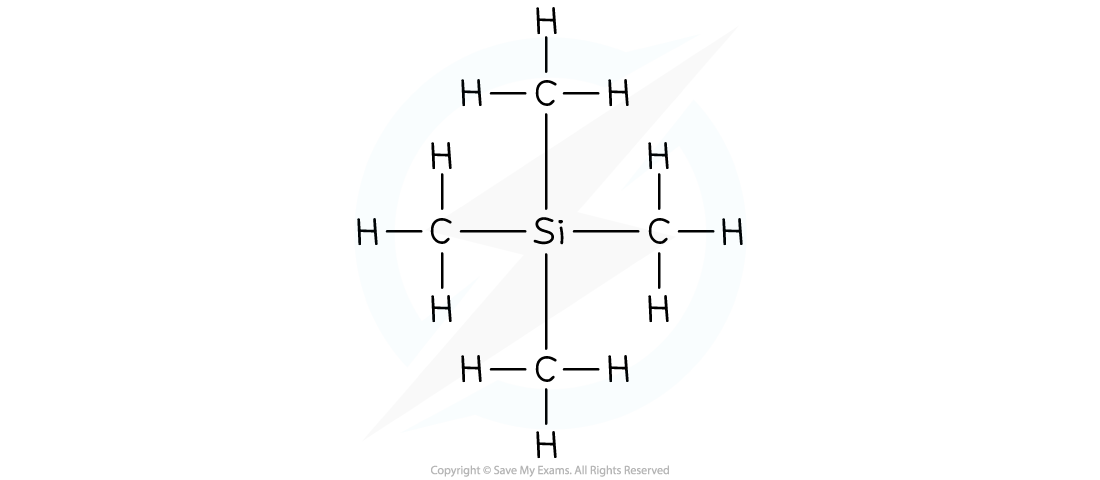 Tetramethylsilane is the common reference compound for NMR spectroscopy
Tetramethylsilane is the common reference compound for NMR spectroscopy
- TMS shows a single sharp peak on NMR spectra, at a value of zero
- TMS is also used because it is:
- Non toxic.
- Does not react with the sample.
- Easily separated from the sample molecule due to its low boiling point.
- Produces one strong, sharp absorption peak on the spectrum.
- Sample peaks are then plotted as a ‘shift’ away from this reference peak
- This gives rise to ‘chemical shift’ values for protons on the sample compound
- Chemical shifts are measured in parts per million (ppm)
Features of a 1H NMR spectrum
- NMR spectra shows the intensity of each peak against their chemical shift
- The area under each peak gives information about the number of protons in a particular environment
- The height of each peak shows the intensity / absorption from protons
- A single sharp peak is seen to the far right of the spectrum
- This is the reference peak from TMS
- Usually at chemical shift 0 ppm
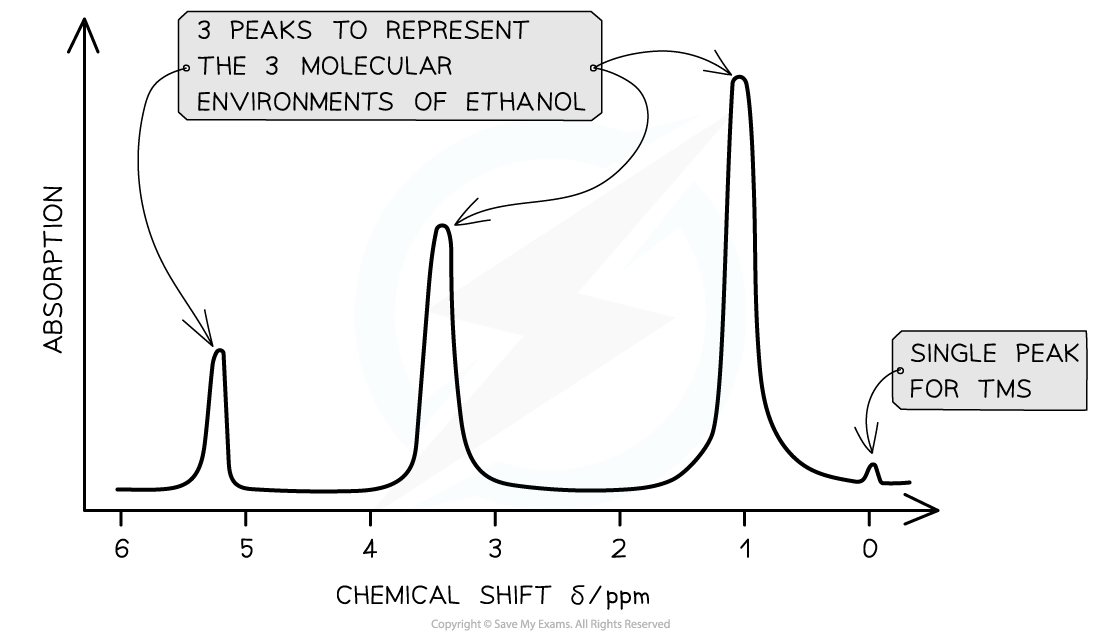
A low resolution 1H NMR for ethanol showing the key features of a spectrum
Molecular environments
- 1H nuclei that have different neighboring atoms (said to have different chemical environments) absorb at slightly different field strengths
- The difference environments are said to cause a chemical shift of the absorption
- Ethanol has the structural formula CH3CH2OH
- There are 3 chemical environments: -CH3, -CH2 and -OH
- The hydrogen atoms in these environments will appear at 3 different chemical shifts
- Different types of protons are given their own range of chemical shifts
Worked Example
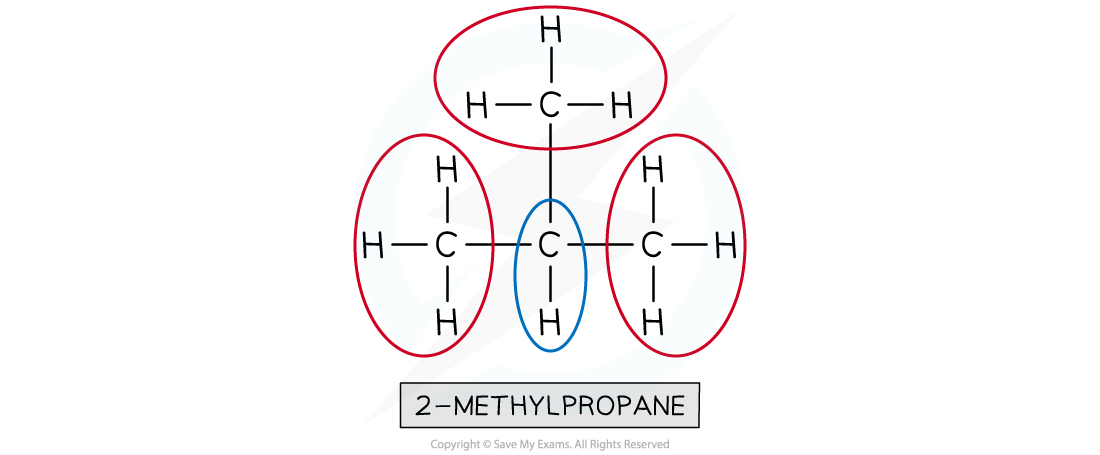
How many different 1H chemical environments occur in 2-methylpropane?
Answer:
Two different 1H chemical environments occur in 2-methylpropane
-
- The three methyl groups are in the same 1H environment
- The lone hydrogen is in its own 1H environment
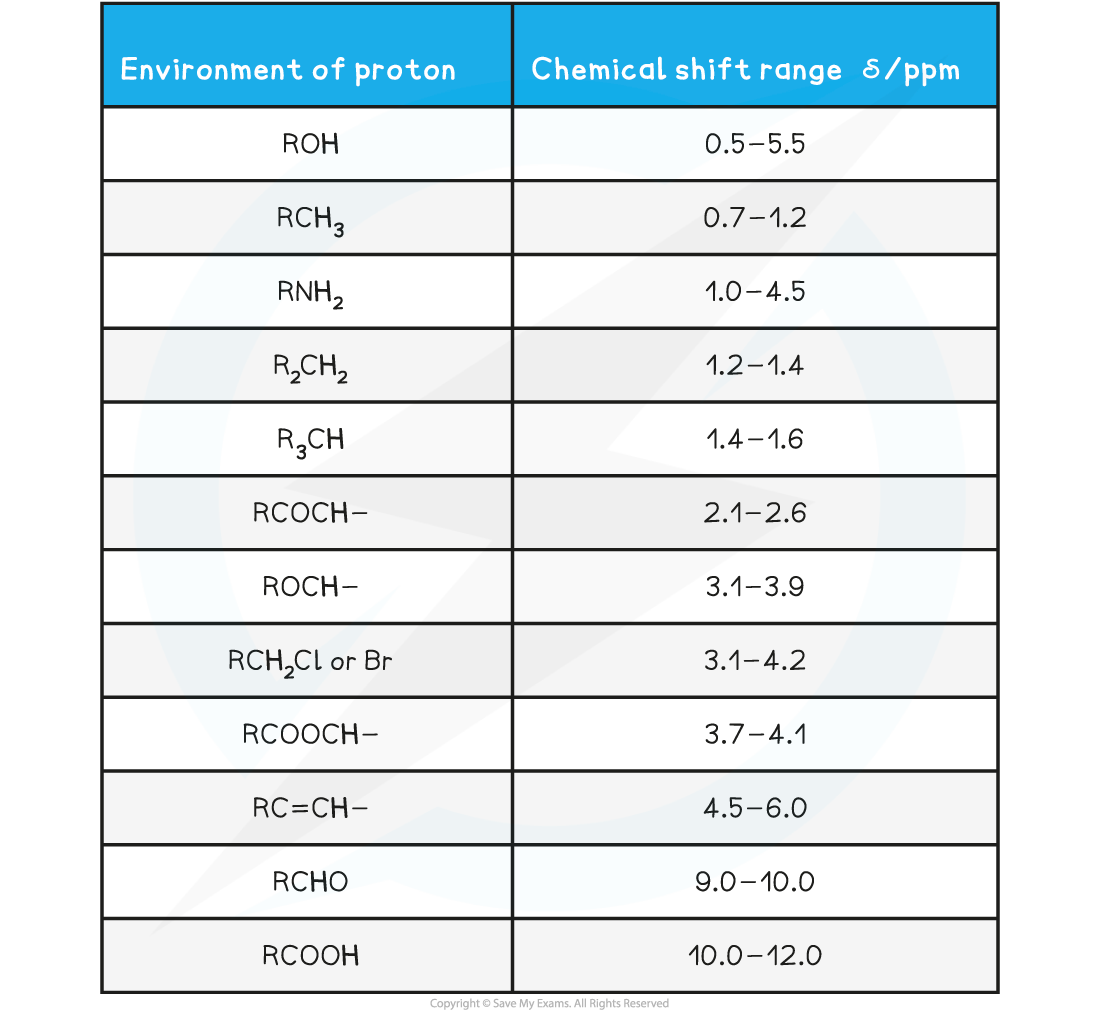
Chemical Shift Values for 1H Molecular Environments Table
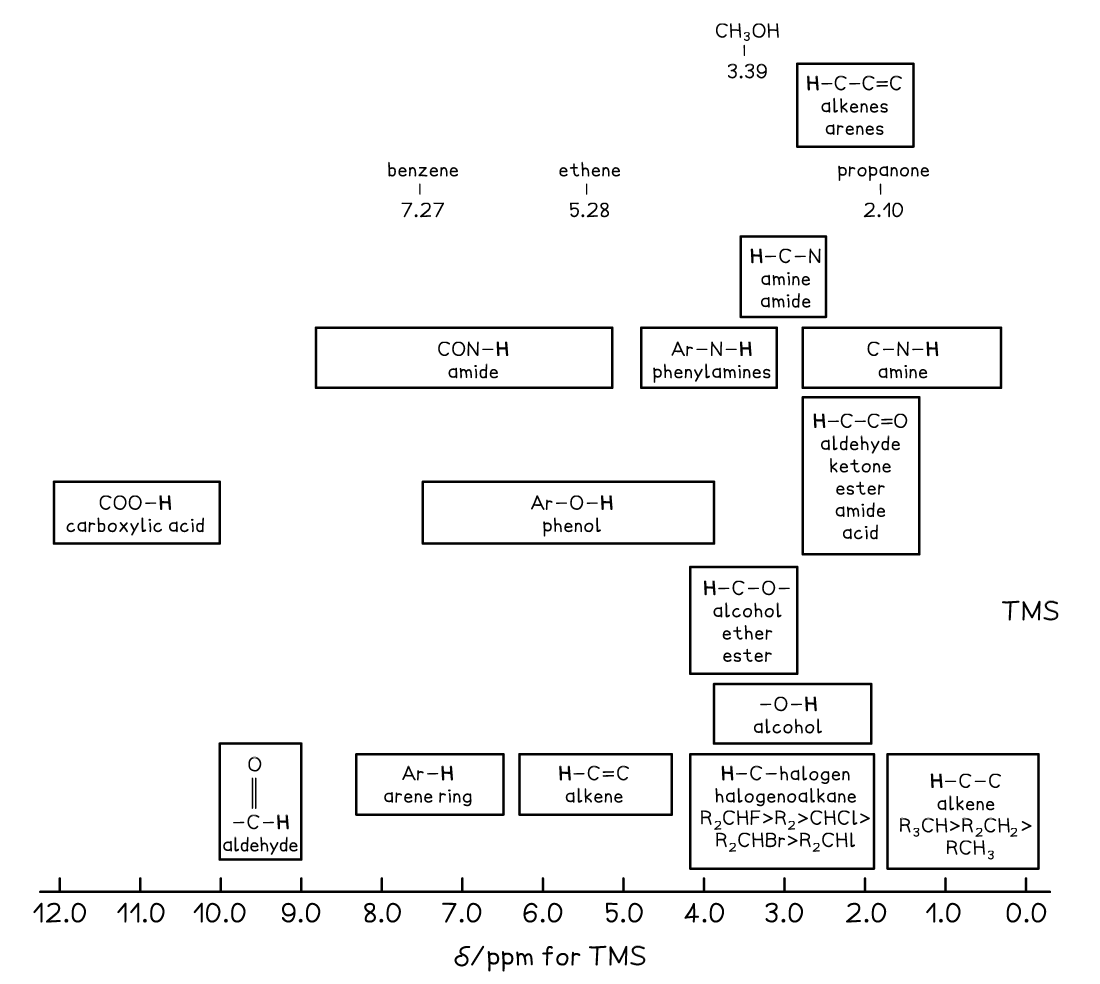
- Protons in the same chemical environment are chemically equivalent
- 1,2-dichloroethane, Cl-CH2-CH2-Cl has one chemical environment as these four hydrogens are all exactly equivalent
- Each individual peak on a 1H NMR spectrum relates to protons in the same environment
- Therefore, 1,2-dichloroethane would produce one single peak on the NMR spectrum as the protons are in the same environment

Low resolution 1H NMR
- Peaks on a low resolution NMR spectrum refers to molecular environments of an organic compound
- Ethanol has the molecular formula CH3CH2OH
- This molecule as 3 separate environments: -CH3, -CH2, -OH
- So 3 peaks would be seen on its spectrum at 1.2 ppm (-CH3), 3.7 ppm (-CH2) and 5.4 ppm (-OH)
- The strengths of the absorptions are proportional to the number of equivalent 1H atoms causing the absorption and are measured by the area underneath each absorption peak
- Hence, the areas of absorptions of -CH3, -CH2, -OH are in the ratio of 3:2:1 respectively
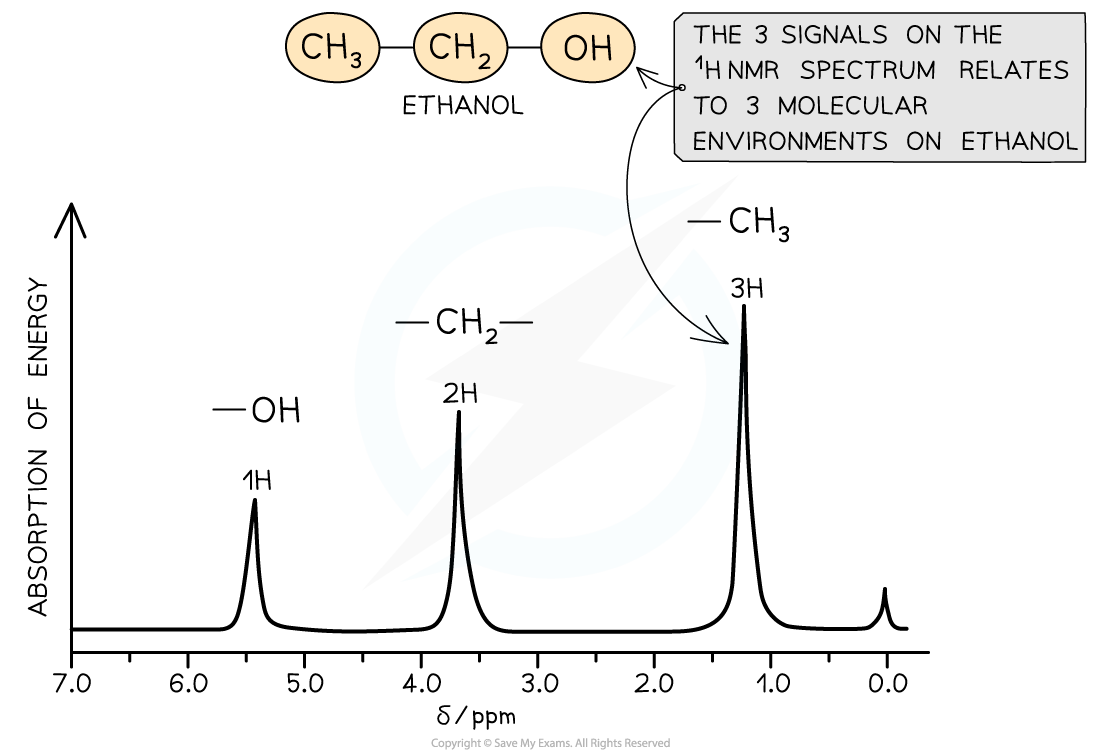
A low resolution NMR spectrum of ethanol showing 3 peaks for the 3 molecular environments
Using High Resolution Proton NMR Data
High resolution 1H NMR
- More structural details can be deduced using high resolution NMR
- The peaks observed on a high resolution NMR may sometimes have smaller peaks clustered together
- The splitting pattern of each peak is determined by the number of protons on neighbouring environments
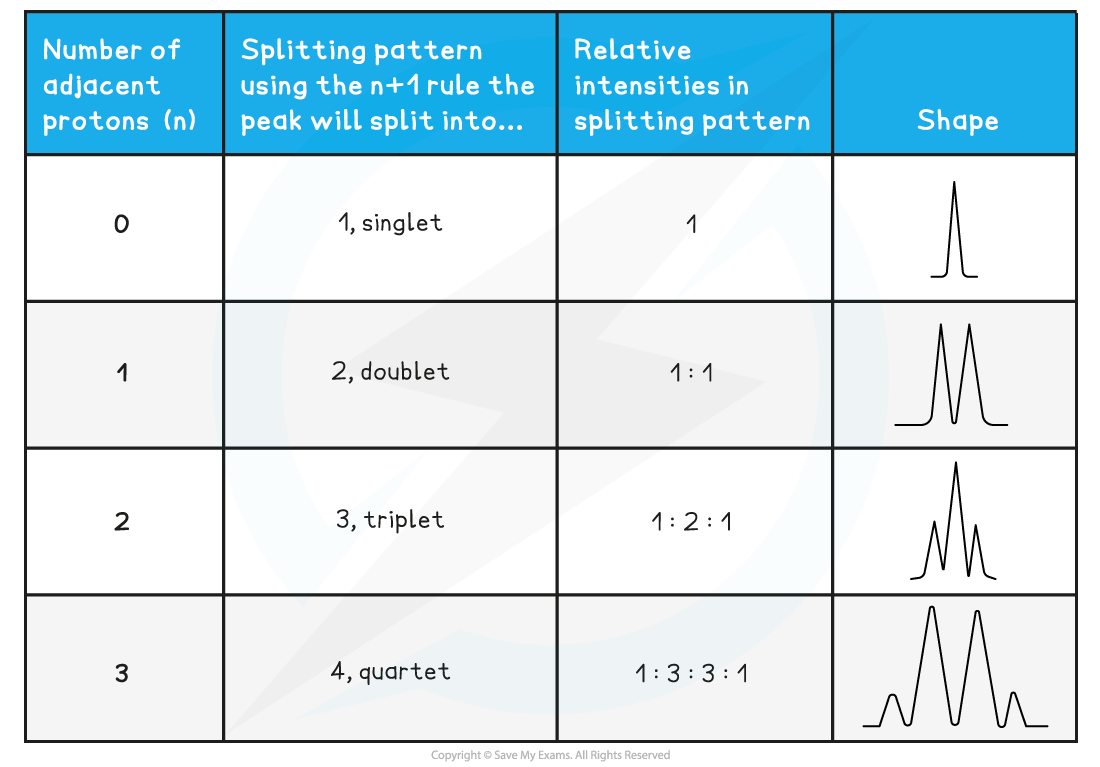
The number of peaks a signal splits into = n + 1
(Where n = the number of protons on the adjacent carbon atom)
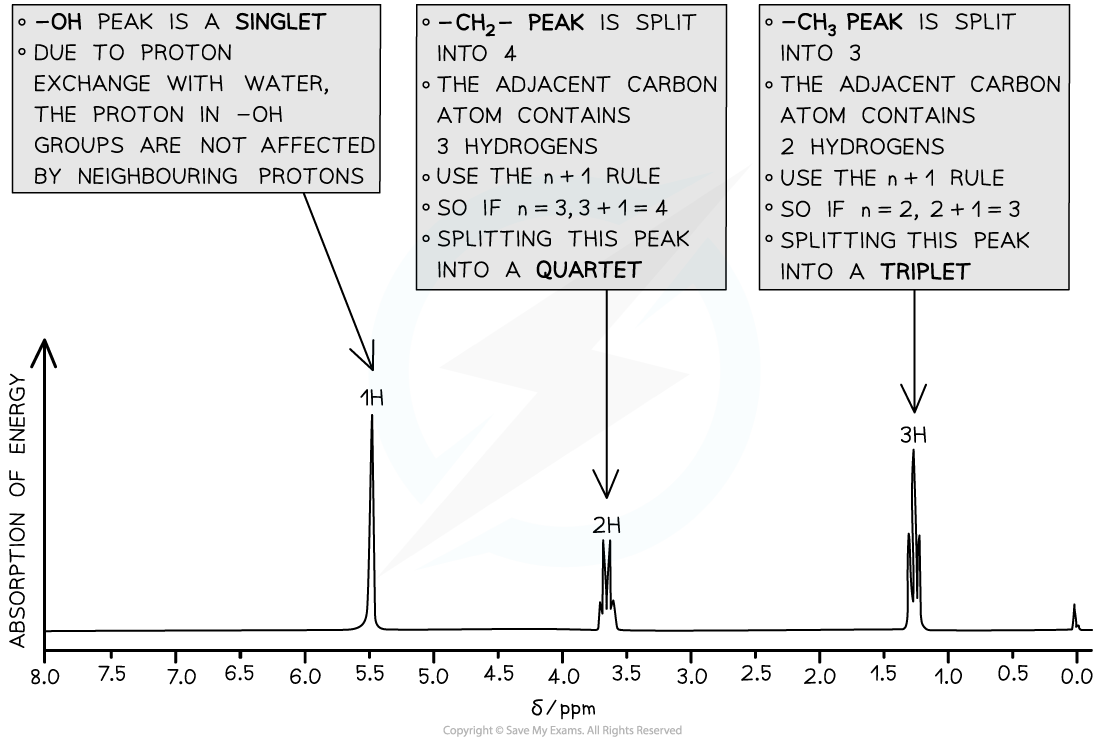
High resolution 1H NMR spectrum of ethanol showing the splitting patterns of each of the 3 peaks. Using the n+1, it is possible to interpret the splitting pattern
- Each splitting pattern also gives information on relative intensities
- A doublet has an intensity ratio of 1:1 – each peak is the same intensity as the other
- In a triplet, the intensity ratio is 1:2:1 – the middle of the peak is twice the intensity of the 2 on either side
- In a quartet, the intensity ratio is 1:2:2:1 – the middle peaks are twice the intensity of the 2 on either side
Integrated spectra
- In 1H NMR, the relative areas under each peak give the ratio of the number of protons responsible for each peak
- The NMR spectrometer measures the area under each peak, as an integration spectra
- This provides invaluable information for identifying an unknown compound
- The 1H NMR of methyl chloroethanoate, ClCH2COOCH3, will show an integration spectra in the peak area ratio of 2:3
- 2 for the protons in the CH2
- 3 for the protons in CH3
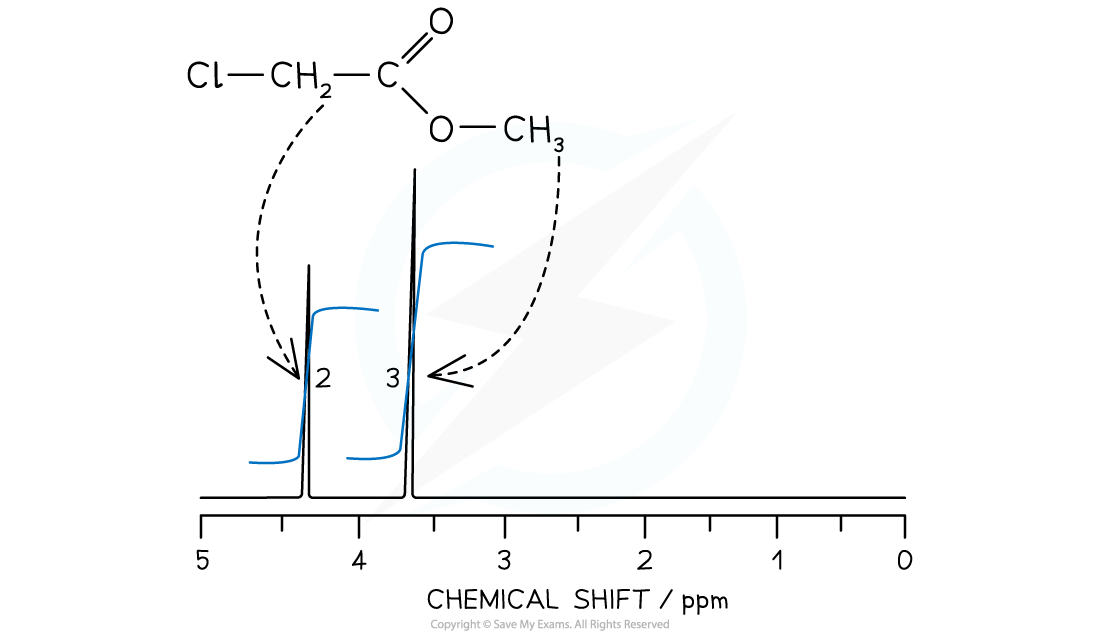
Spin-Spin Splitting
- A 1H NMR peak can show you the structure of the molecule but also the peaks can be split into sub-peaks or splitting patterns
- These are caused by a proton's spin interacting with the spin states of nearby protons that are in different environments
- This can provide information about the number of protons bonded to adjacent carbon atoms
- The splitting of a main peak into sub-peaks is called spin-spin splitting
The n+1 rule
- The number of sub-peaks is one greater than the number of adjacent protons causing the splitting
- For a proton with n protons attached to an adjacent carbon atom, the number of sub-peaks in a splitting pattern = n+1
- When analysing spin-spin splitting, it shows you the number of hydrogen atoms on the immediately adjacent carbon atom
- These are the splitting patterns that you need to be able to recognise from a 1H spectra:
1H NMR Peak Splitting Patterns Table
- Splitting patterns must occur in pairs, because each protons splits the signal of the other
- There are some common splitting pairs you will see in a spectrum however you don't need to learn these but can be worked out using the n+1 rule
- You will quickly come to recognise the triplet / quartet combination for a CH3CH2 because it is so common
Common pair of splitting patterns
- A quartet and a triplet in the same spectrum usually indicate an ethyl group, CH3CH2-
- The signal from the CH3 protons is split as a triplet by having two neighbours
- The signal from the CH2 protons is split as a quartet by having three neighbours
- Here are some more common pairs of splitting patterns

Common pairs of splitting patterns
1H NMR spectrum of propane
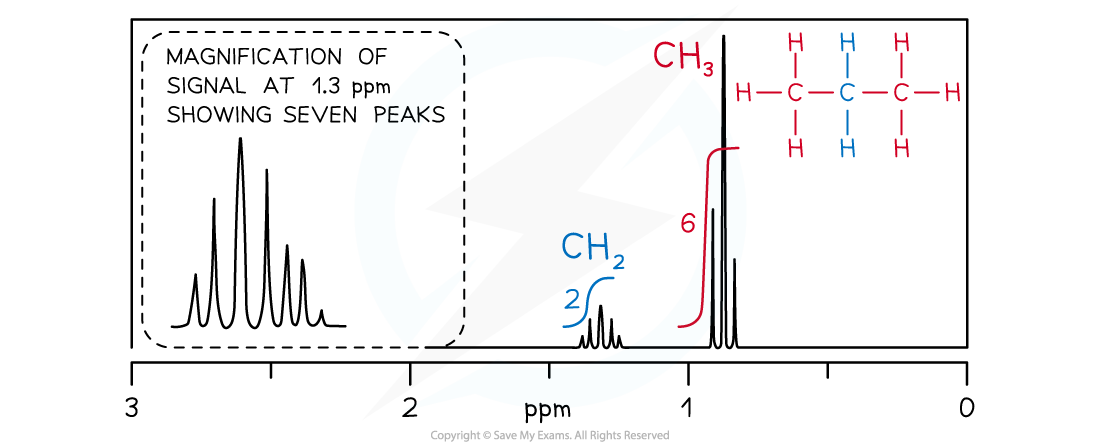
- The CH2 signal in propane (blue) is observed as a heptet because it has six neighbouring equivalent H atoms (n+1 rule), three either side in two equivalent CH3 groups
- The CH3 groups (red) produce identical triplets by coupling with the CH2 group
Worked Example
For the compound (CH3)2CHOH predict the following:
i) the number of peaks
ii) the type of proton and chemical shift (using the Data sheet)
iii) the relative peak areas
iv) the split pattern
Answers:
i) 3 peaks
ii) (CH3)2CHOH at 0.7 - 1.2 ppm, (CH3)2CHOH at 3.1 - 3.9 ppm, (CH3)2CHOH at 0.5 - 5.5 ppm
iii) Ratio 6 : 1 : 1
iv) (CH3)2CHOH split into a doublet (1+1=2), (CH3)2CHOH split into a heptet (6+1=7)
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





