- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A Level Chemistry:复习笔记3.5.3 Organic Liquid Preparation & Purification
Organic Liquid Preparation & Purification
Simple Distillation
- This is used to separate a liquid and soluble solid from a solution (e.g. water from a solution of salt water) or a pure liquid from a mixture of liquids
- The solution is heated, and pure water evaporates producing a vapour which rises through the neck of the round bottomed flask
- The vapour passes through the condenser, where it cools and condenses, turning into the pure liquid that is collected in a beaker
- After all the water is evaporated from the solution, only the solid solute will be left behind

Diagram showing the distillation of a mixture of salt and water
Fractional Distillation
- This is used to separate two or more liquids that are miscible with one another (e.g., ethanol and water from a mixture of the two)
- The solution is heated to the temperature of the substance with the lowest boiling point
- This substance will rise and evaporate first, and vapours will pass through a condenser, where they cool and condense, turning into a liquid that will be collected in a beaker
- All of the substance is evaporated and collected, leaving behind the other components(s) of the mixture
- For water and ethanol
- Ethanol has a boiling point of 78 ºC and water of 100 ºC
- The mixture is heated until it reaches 78 ºC, at which point the ethanol boils and distills out of the mixture and condenses into the beaker
- When the temperature starts to increase to 100 ºC heating should be stopped. Water and ethanol are now separated
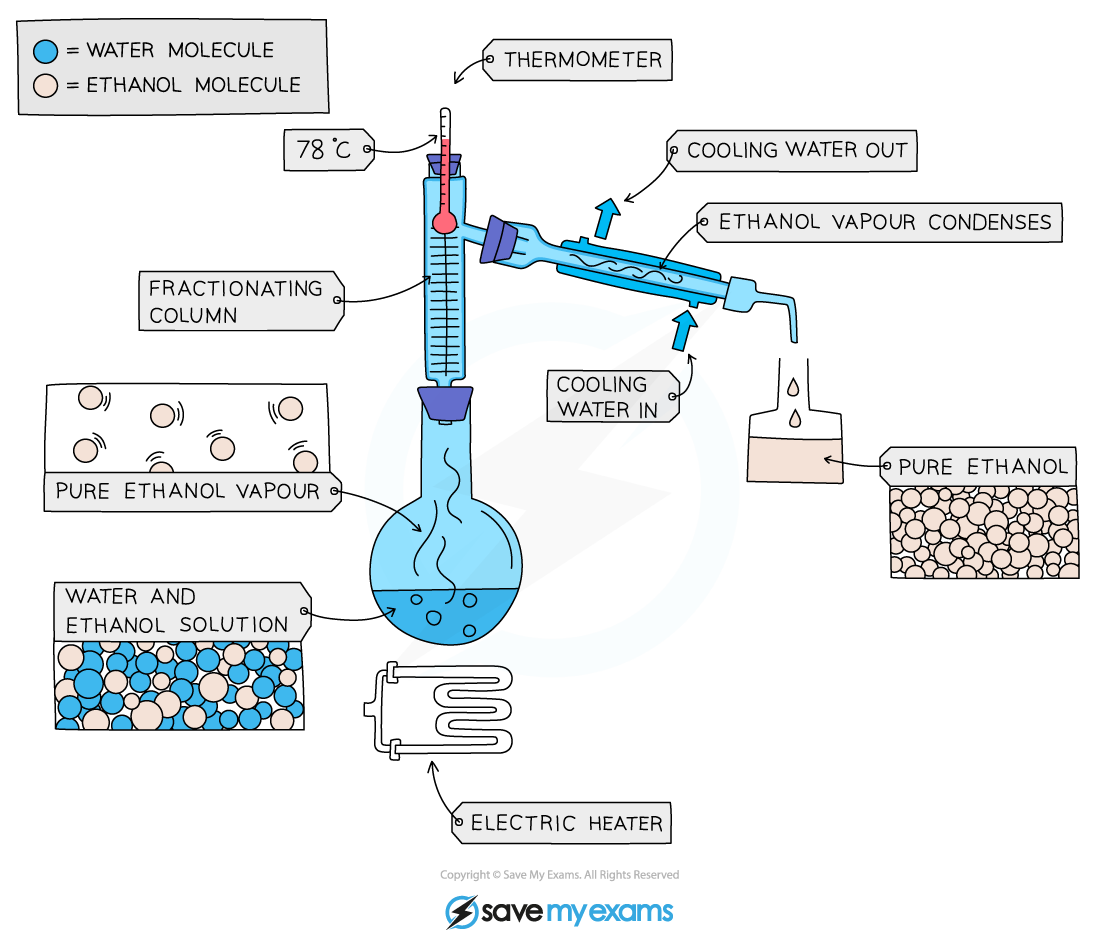
Fractional distillation of a mixture of ethanol and water
Solvent Extraction
- This method uses a solvent to remove a desired organic product from a reaction mixture
- Various solvents can be used but the solvent should have the following features:
- Immiscible (does not mix) with the solvent containing the desired organic product
- The desired organic product should be much more soluble than the added solvent
- The process is summarised as follows
- Place the reaction mixture in a separating funnel and add the chosen solvent forming a separate layer
- Place a stopper in the neck of the funnel and gently shake the contents of the funnel for a while
- Allow the contents of the funnel to settle into two layers
- Remove the stopper and open the tap to allow the lower layer to drain into a flask
- Pour the remaining layer into a separate flask
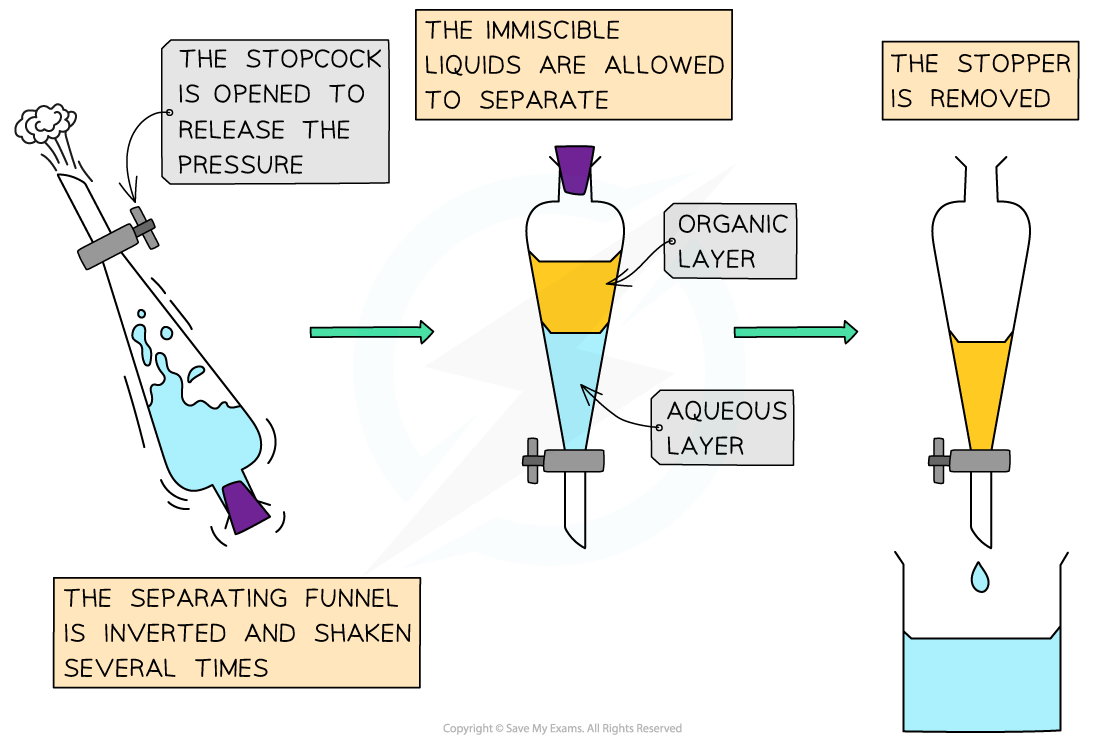
Process of solvent extraction
- Using smaller volumes of solvent in stages is more efficient at removing the desired organic product compared to using the same volume in one portion
Crystallisation
- Used to separate a dissolved solid from a solution, when the solid is much more soluble in hot solvent than in cold (e.g., copper sulphate from a solution of copper (II) sulphate in water)
- The solution is heated, allowing the solvent to evaporate, leaving a saturated solution behind
- Test if the solution is saturated by dipping a clean, dry, cold glass rod into the solution
- If the solution is saturated, crystals will form on the glass rod
- The saturated solution is allowed to cool slowly
- Crystals begin to grow as solids will come out of solution due to decreasing solubility
- The crystals are collected by filtering the solution, they are washed with cold distilled water to remove impurities and are then allowed to dry
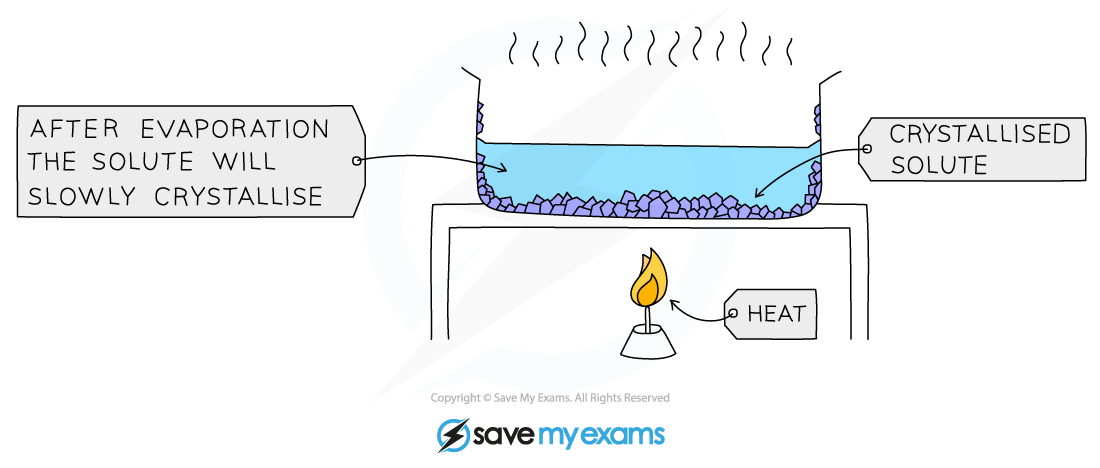
Diagram showing the process of crystallisation
Boiling point determination
- A liquids purity can be determined by measuring its boiling point
- Impurities present in the liquid will raise its boiling temperature
- The boiling points of pure organic compounds have been carefully measured and are widely available in data books and online
- The measured boiling point can be compared to the data book value to determine how pure the compound is
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





