- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A Level Chemistry:复习笔记3.3.7 Waste Polymers
Disposal of Polymers
- Polymers provide a readily available, cheap alternative to many metal, glass, paper and cardboard materials in everyday use
- The low reactivity of many polymers makes them ideal for certain uses, e.g. food packaging, but at the same time creates problems with their disposal as a lot of polymers are non-biodegradable
- There are many published articles about the environmental problems of waste plastic killing marine animals
- One method of polymer disposal continues to be the use of landfill sites
- This is not ideal and various initiatives are being introduced aiming to reduce this method of waste disposal in general as well as with specific regard to polymers
Recycling
- Polymer recycling reduces the amount of waste that it going to landfill sites
- Newer landfill sites can have a recycling point where the new waste is brought before going to into the actual landfill - this is in an effort to reduce the amount of polymers (and other recyclable materials) unnecessarily going into the waste site
- The recycling of polymers can also reduce the use of finite resources
- Lots of polymers are made from the products of cracking crude oil and it's fractions
- Recycling polymers is a time-consuming process as they have to be sorted into the different categories
- These categories are usually shown somewhere on the plastic / polymer product with the recycling symbol and numbers or abbreviations for the different polymers, e.g.:

The recycling symbol for the polyethylene terephthalate polymer
- After sorting, the polymers are chopped, washed, dried, melted and then cast into pellets ready for use
- However, mixed polymers can mean that this process is wasted as its produces an unusable mix of polymers
- Certain polymers can cause problems when recycling due to their chemical composition, e.g. PVC contains a large amount of toxic chlorine which can be released
- Modern techniques are overcoming this PVC problem by dissolving the polymer and precipitating out the recycled material
Incineration
- Some petroleum / natural gas derived polymers are still difficult to recycle
- Since they have a large amount of energy stored within the polymer chains, these polymers can be incinerated
- This process can then be used to boil water and use the water vapour to turn turbines inside a power station, in a similar fashion to coal-fired power stations
- This process still causes environmental pollution as the carbon within the polymer can be released as carbon dioxide contributing to global warming
- Other toxic waste products include hydrogen chloride from the combustion of PVC
Feedstock recycling
- Feedstock recycling is where waste polymers are broken down, by chemical and thermal processes, into monomers, gases and oils
- These products are then used as the raw materials in the production of new polymers and other organic chemicals
- The major benefit of feedstock recycling, compared to other methods of polymer disposal, is that it works with unsorted and unwashed polymers
Sustainability
- Chemists will often use the principles of green chemistry when designing a sustainable polymer manufacturing process
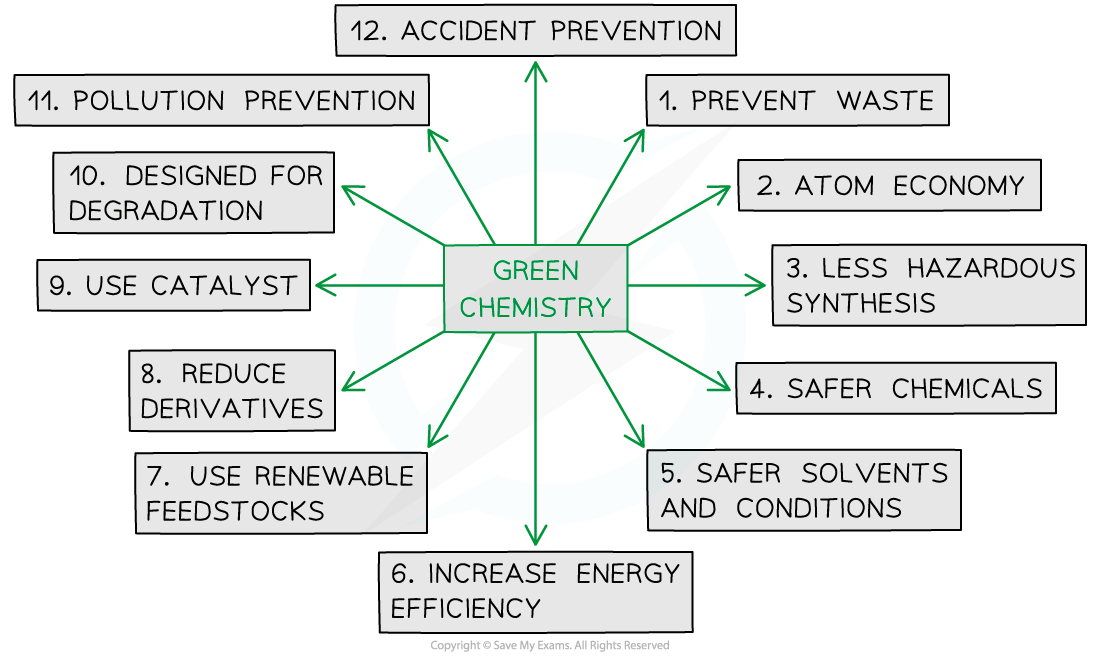
The twelve principles of green chemistry
- They will aim to:
- Use chemicals (reactants, solvents and catalysts) that are as safe and environmentally friendly as possible
- They also aim to reduce the amount of chemicals used, in terms of physical quantities as well as actual number of chemicals
- Use renewable feedstock chemicals where possible
- Reduce the energy requirements for the reaction as well as increase the energy efficiency, which has both an environmental and financial bonus
- Improve atom economy / reduce the amount of waste byproducts
- Consider the lifespan of the polymer, which will incorporate the idea of the polymer being suitable for its use
- Use chemicals (reactants, solvents and catalysts) that are as safe and environmentally friendly as possible
Limiting Problems of Polymer Disposal
- Chemists have designed ways to remove toxic waste products like HCl before they are emitted into the atmosphere
- The waste gase from the incinerator are scrubbed/reacted with a base or carbonate
- The base reacts with the acidic HCl gas, neutralising it
- eg. CaO (s) + 2HCl (aq)→ CaCl2 (aq) + H2O (l)
- Chemists have also developed biodegradable and compostable polymers
Biodegradable polymers
- Biodegradable polymers can be broken down over time by microorganisms
- Common products from this process include carbon dioxide, water and other organic compounds
- The polyester and polyamide condensation polymers are considered to be biodegradable as they can be broken down using hydrolysis reactions
- This is a major advantage over the polymers produced using alkene monomers (polyalkenes)
- When polyesters and polyamides are taken to landfill sites, they can be broken down easily and their products used for other applications
Compostable polymers
- Compostable polymers are commonly plant based
- Plant starch is being used in the production of biodegradable bin liners
- Sugar cane fibres are replacing polystyrene in the production of disposable plates and cups
- Compostable polymers degrade naturally leaving no harmful residues
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





