- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A Level Chemistry:复习笔记3.3.3 Electrophilic Addition
Electrophilic Addition
- The double bond in alkenes is an area of high electron density (there are four electrons found in this double bond)
- This makes the double bond susceptible to attack by electrophiles (electron-loving species)
- An electrophilic addition is the addition of an electrophile to a double bond
- The C-C double bond is broken, and a new single bond is formed from each of the two carbon atoms
- Electrophilic addition reactions include the addition of:
- Hydrogen (also known as hydrogenation reaction)
- Steam (H2O (g))
- Hydrogen halide (HX)
- Halogen

The diagram shows an overview of the different electrophilic addition reactions alkenes can undergo
Manufacture of margarine
- Hydrogenation is extensively used in industry to manufacture margarine
- Naturally occurring vegetable oils are unsaturated and contain C=C double bonds
- When these bonds react with hydrogen, some of the C=C double bonds become C-C single bonds
- This process changes the properties of vegetable oil and converts it into a solid: margarine
Oxidation
- Alkenes can also be oxidised by acidified potassium manganate(VII) (KMnO4) which is a very powerful oxidising agent
- When shaken with cold dilute KMnO4 the pale purple solution turns colourless and the product is a diol
- This colour change means this reaction can be used, like bromine, to distinguish alkanes from alkenes ( alkanes do not have double bonds and so are not oxidised in this way)
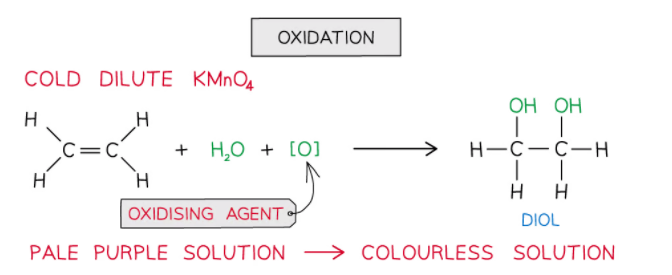
- Although you do not need to know the full details of the working of this reaction you can think of it as an oxidation followed by an addition
- The potassium manganate provides an oxygen atom (oxidation)
- Then water in the solution provides another oxygen atom and two hydrogen atoms, so there is addition of two OH groups across the double bond
Heterolytic Fission
- Heterolytic fission is breaking a covalent bond in such a way that the more electronegative atom takes both the electrons from the bond to form a negative ion and leaving behind a positive ion
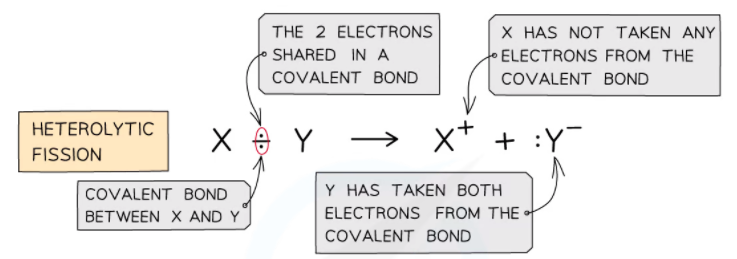
The diagram shows heterolytic fission in which the most electronegative atom takes both electrons in the covalent bond
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





