- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A Level Chemistry:复习笔记1.5.4 Intermolecular Forces & Physical Properties
Intermolecular Forces & Physical Properties
Branching
- The larger the surface area of a molecule, the more contact it will have with adjacent molecules
- The surface area of a molecule is reduced by branching
- The greater its ability to induce a dipole in an adjacent molecule, the greater the London (dispersion) forces and the higher the melting and boiling points
- This point can be illustrated by comparing different isomers containing the same number of electrons:
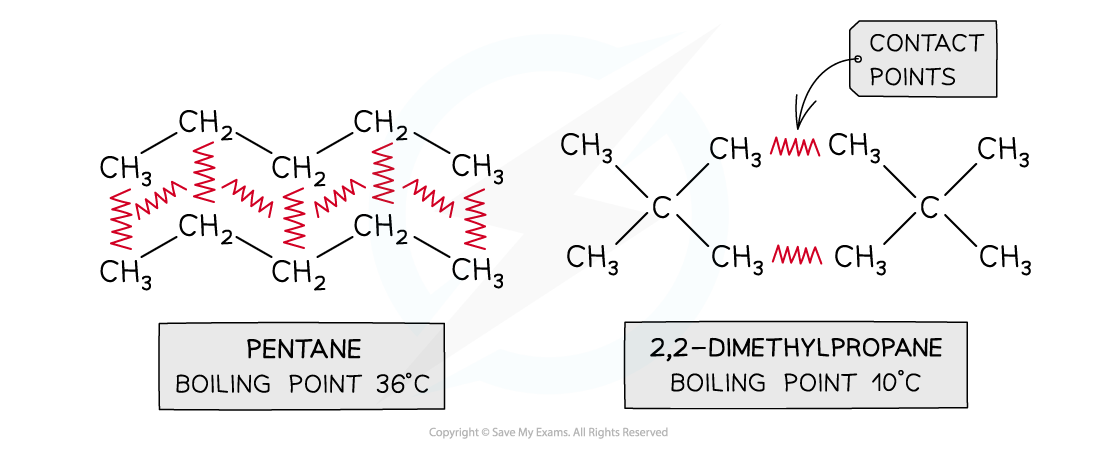
Boiling points of molecules with the same numbers of electrons but different surface areas
Number of electrons
- The greater the number of electrons (or the greater the molecular mass) in a molecule, the greater the likelihood of a distortion and thus the greater the frequency and magnitude of the temporary dipoles
- The dispersion forces between the molecules are stronger and the enthalpy of vaporisation, melting and boiling points are larger
- The greater boiling points of the noble gases illustrate this factor:
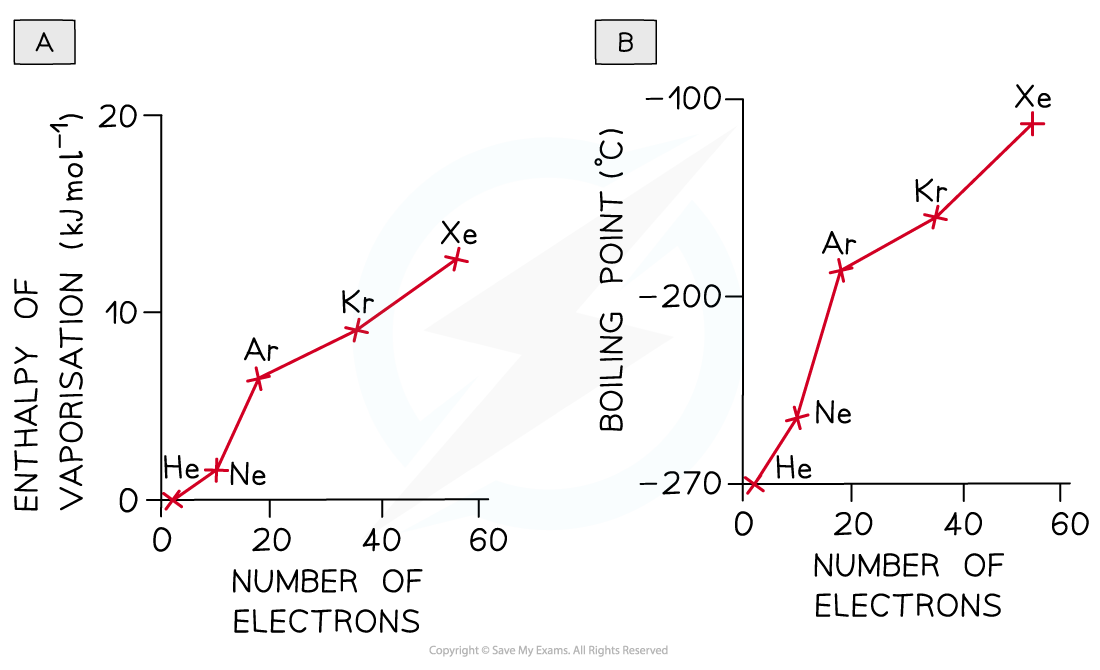
As the number of electrons increases more energy is needed to overcome the forces of attraction between the noble gases atoms
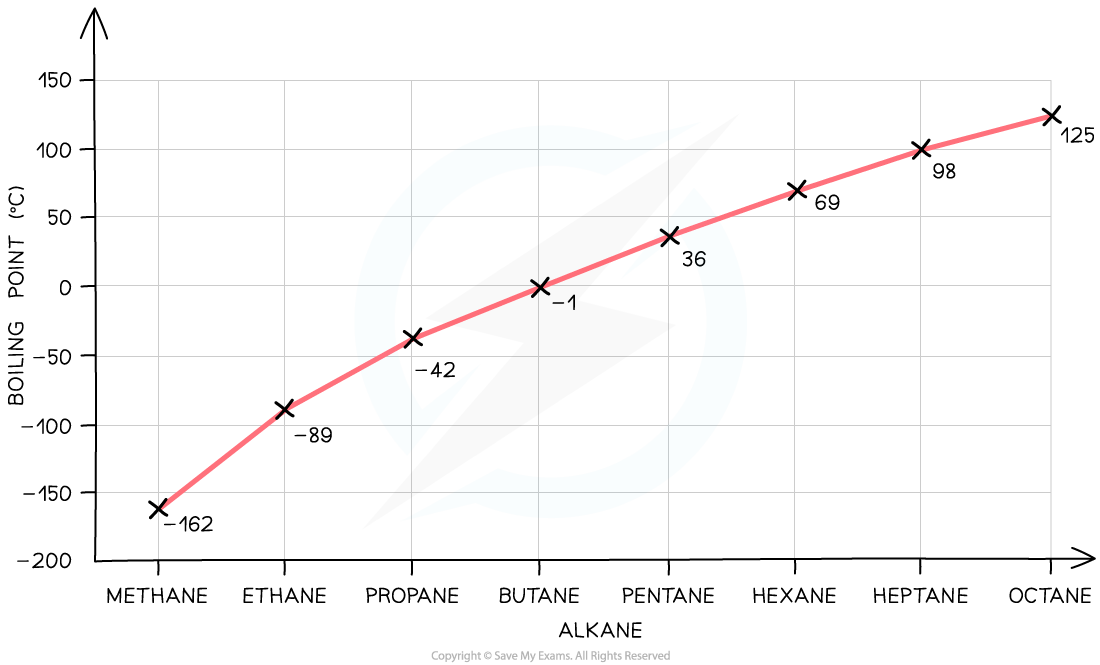
Graph showing the increase in boiling point as the number of electrons increases
Alcohols
- Hydrogen bonding occurs between molecules where you have a hydrogen atom attached to one of the very electronegative elements - fluorine, oxygen or nitrogen
- In an alcohol, there are O-H bonds present in the structure
- Therefore hydrogen bonds set up between the slightly positive hydrogen atoms (δ+ H) and lone pairs on oxygens in other molecules
- The hydrogen atoms are slightly positive because the bonding electrons are pulled away from them towards the very electronegative oxygen atoms
- In alkanes, the only intermolecular forces are temporary induced dipole-dipole forces
- Hydrogen bonds are much stronger than these and therefore it takes more energy to separate alcohol molecules than it does to separate alkane molecules
- Therefore, the boiling point of alkanes is lower than the boiling point of the respective alcohols
- For example, the boiling point of propane is -42 oC and the boiling point of propanol is 97 oC
Hydrogen Halides
- The boiling points of the hydrogen halides are as follows
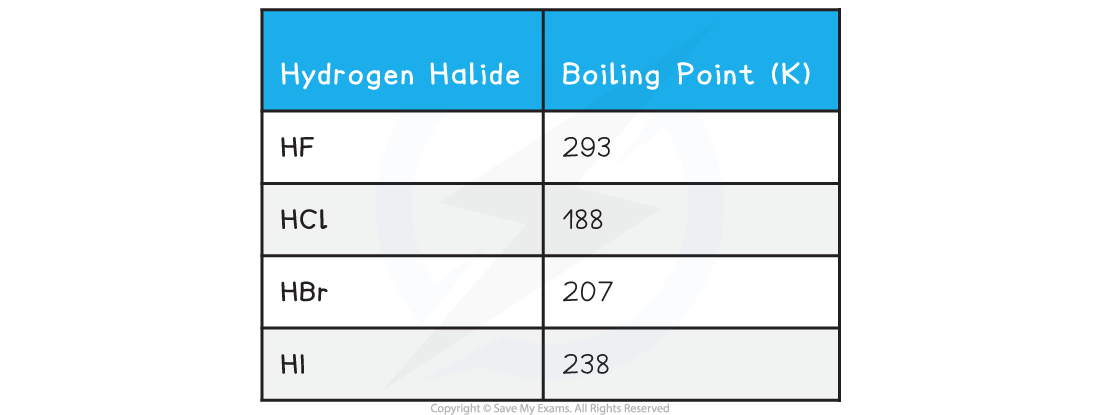
- The boiling points of the rest of the hydrogen halides increase as the molecules become larger
- The extra electrons allow greater temporary dipoles and so increase the amount of London dispersion forces between the molecules
- Hydrogen fluoride also has hydrogen bonding between the HF molecules
- The bond is very polar so that the hydrogen has a significant amount of positive charge and the fluorine a significant amount of negative charge. In addition, the fluorine has small intense lone pairs
- Hydrogen bonds can form between the hydrogen on one molecule and a lone pair on the fluorine in its neighbour
Choosing Solvents
Solubility
- The general principle is that 'like dissolves like' so non-polar substances mostly dissolve in non-polar solvents, like hydrocarbons and they form dispersion forces between the solvent and the solute
- Polar covalent substances generally dissolve in polar solvents as a result of dipole-dipole interactions or the formation of hydrogen bonds between the solute and the solvent
- A good example of this is seen in organic molecules such as alcohols and water:
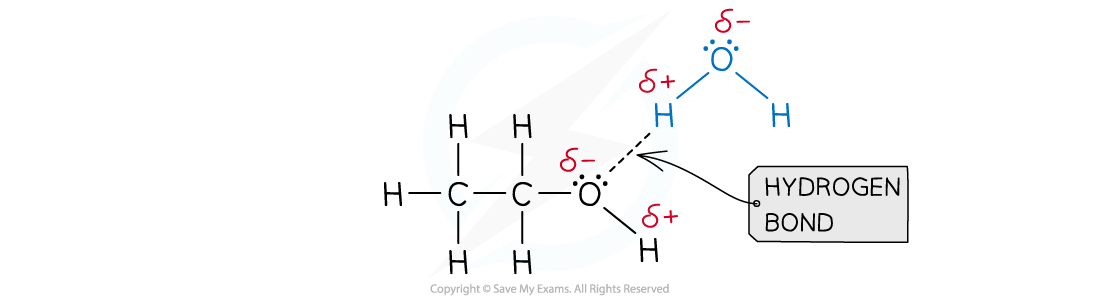
Hydrogen bonds form between ethanol and water
- As covalent molecules become larger their solubility can decrease as the polar part of the molecule is only a smaller part of the overall structure
- This effect is seen in alcohols for example where ethanol, C2H5OH, is readily soluble but hexanol, C6H13OH, is not
- Polar covalent substances are unable to dissolve well in non-polar solvents as their dipole-dipole attractions are unable to interact well with the solvent
- Giant covalent substances generally don't dissolve in any solvents as the energy needed to overcome the strong covalent bonds in the lattice structures is too great
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





