- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A Level Chemistry:复习笔记1.5.3 Hydrogen Bonding
Hydrogen Bonding Interactions
Examples of compounds that can form hydrogen bonds are:
- Alcohols (contains an O-H bond)
- Ammonia (contains an N-H bond)
- Amines (contains an N-H bond)
- Carboxylic acids (contains an O-H bond)
- Hydrogen fluoride (contains an H-F bond)
- Proteins (contains an N-H bond)
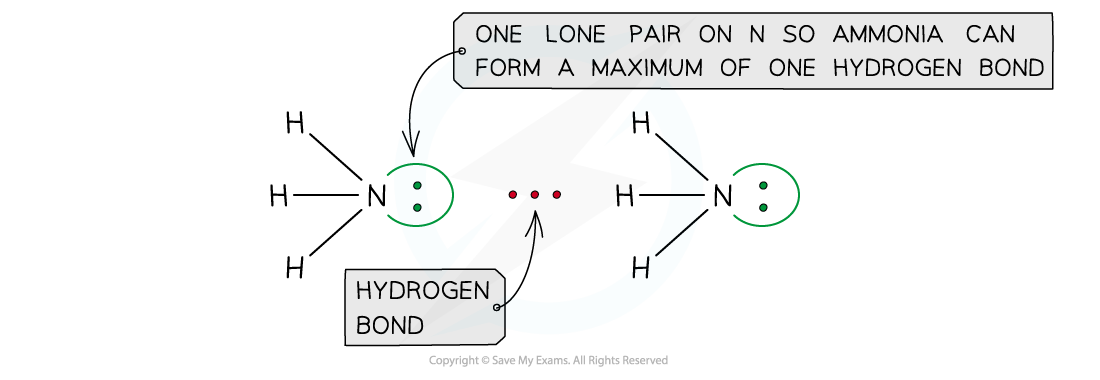
Ammonia can form a maximum of one hydrogen bond per molecule
Anomalous Properties of Water
Properties of water
- Hydrogen bonding in water, causes it to have anomalous properties such as high melting and boiling points, high surface tension and a higher density in the liquid than the solid
High melting & boiling points
- Water has high melting and boiling points due to the the strong intermolecular forces of hydrogen bonding between the molecules in both ice (solid H2O) and water (liquid H2O)
- A lot of energy is therefore required to separate the water molecules and melt or boil them
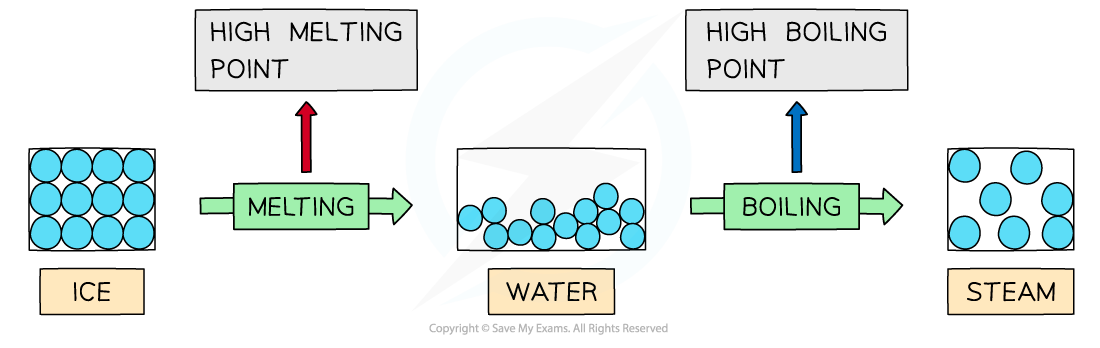
Hydrogen bonds are strong intermolecular forces which are harder to break causing water to have a higher melting and boiling point than would be expected for a molecule of such a small size
- The graph below compares the enthalpy of vaporisation (energy required to boil a substance) of different hydrides
- The enthalpy changes increase going from H2S to H2Te due to the increased number of electrons in the Group 16 elements
- This causes an increase in the instantaneous dipole - induced dipole forces (dispersion forces) as the molecules become larger
- Based on this, H2O should have a much lower enthalpy change (around 17 kJ mol-1)
- However, the enthalpy change of vaporisation is almost 3 times larger which is caused by the hydrogen bonds present in water but not in the other hydrides
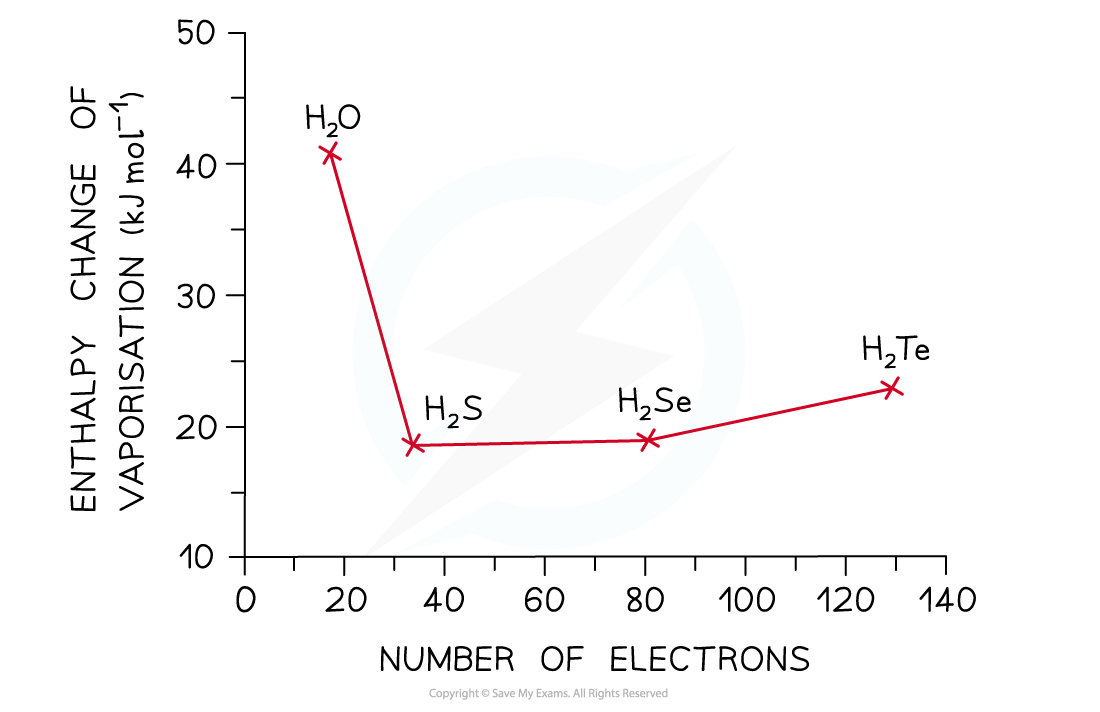
The high enthalpy change of evaporation of water suggests that instantaneous dipole-induced dipole forces are not the only forces present in the molecule – there are also strong hydrogen bonds, which cause the high boiling point
High surface tension
- Water has a high surface tension
- Surface tension is the ability of a liquid surface to resist any external forces (i.e. to stay unaffected by forces acting on the surface)
- The water molecules at the surface of liquid are bonded to other water molecules through hydrogen bonds
- These molecules pull downwards the surface molecules causing the surface of them to become compressed and more tightly together at the surface
- This increases water’s surface tension
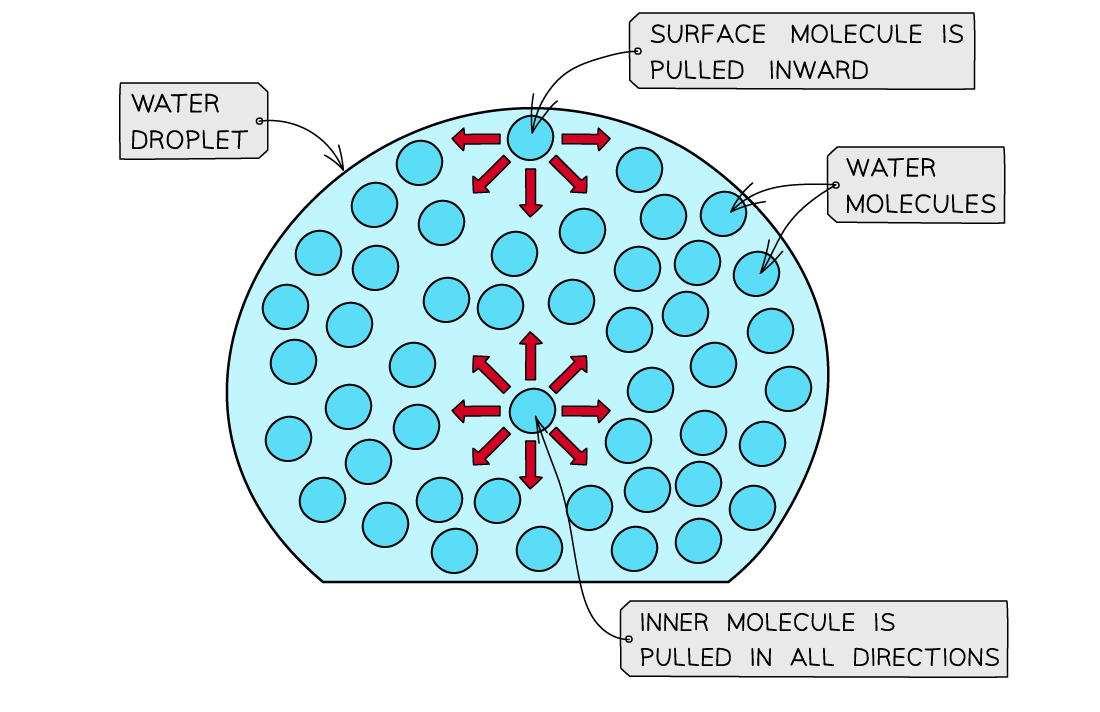
The surface molecules are pulled downwards due to the hydrogen bonds with other molecules, whereas the inner water molecules are pulled in all directions
Density
- Solids are denser than their liquids as the particles in solids are more closely packed together than in their liquid state
- The water molecules are packed into an open lattice
- This way of packing the molecules and the relatively long bond lengths of the hydrogen bonds means that the water molecules are slightly further apart than in the liquid form
- Therefore, ice has a lower density than liquid water by about 9%
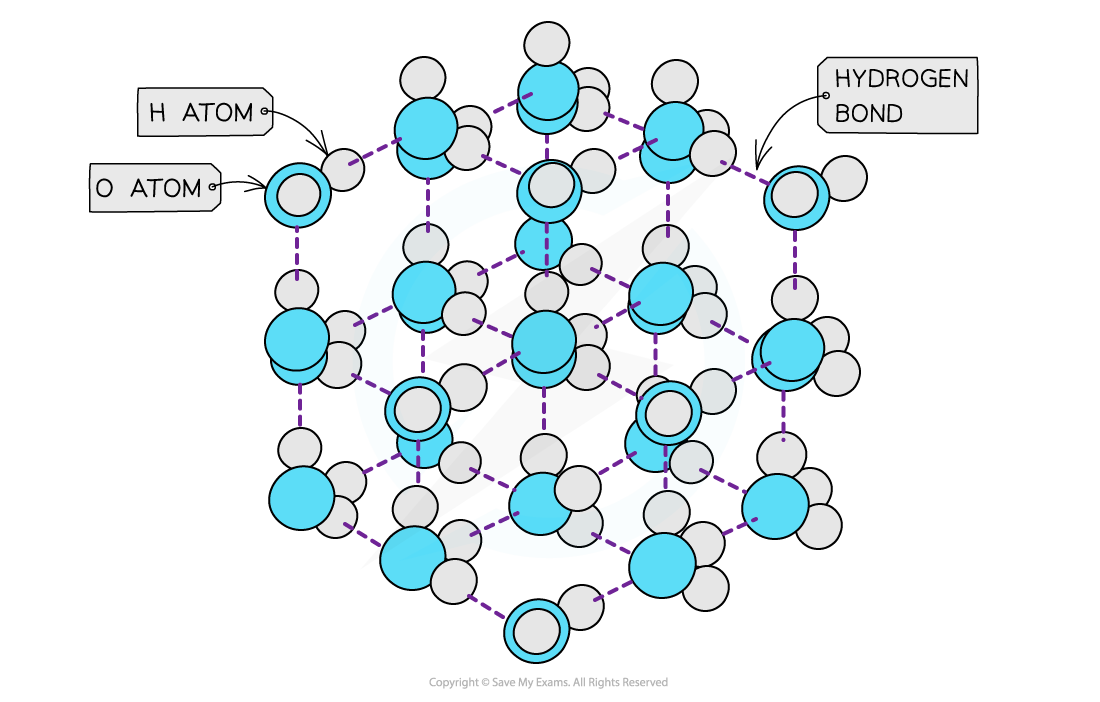
The ‘more open’ structure of molecules in ice causes it to have a lower density than liquid water
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





