- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
CIE A Level Chemistry复习笔记4.1.2 Mass Spectrometry
Interpreting Mass Spectra
- Mass spectroscopy is an analytical technique used to identify unknown compounds
- The molecules in the small sample are bombarded with high energy electrons which can cause the molecule to lose an electron
- This results in the formation of a positively charged molecular ion with one unpaired electron
- One of the electrons in the pair has been removed by the beam of electrons

- The molecular ion can further fragment to form new ions, molecules, and radicals
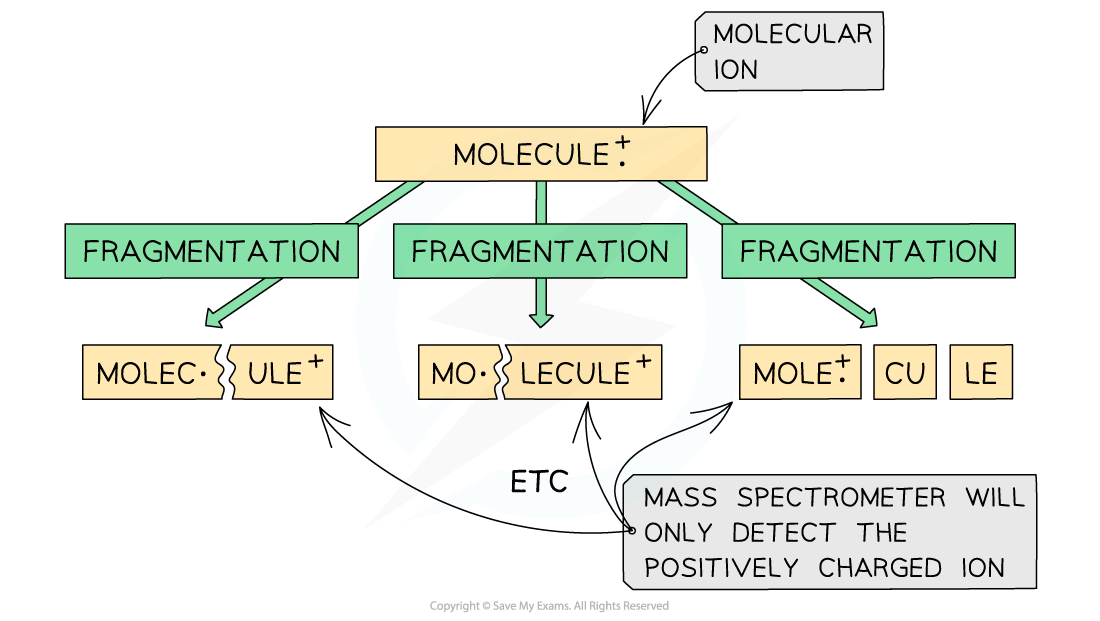
Fragmentation of a molecule in mass spectroscopy
- These fragmentation ions are accelerated by an electric field
- Based on their mass (m) to charge (e) ratio, the fragments of ions are then separated by deflecting them into the detector
- For example, an ion with mass 16 and charge 2+ will have a m/e value of 8
- The smaller and more positively charged fragment ions will be detected first as they will get deflected the most and are more attracted to the negative pole of the magnet
- Each fragment corresponds to a specific peak with a particular m/e value in the mass spectrum
- The base peak is the peak corresponding to the most abundant ion
Isotopes
- Isotopes are different atoms of the same element that contain the same number of protons and electrons but a different number of neutrons.
- These are atoms of the same elements but with different mass number
- For example, Cl-35 and Cl-37 are isotopes as they are both atoms of the same element (chlorine, Cl) but have a different mass number (35 and 37 respectively)
- Mass spectroscopy can be used to find the relative abundance of the isotopes experimentally
- The relative abundance of an isotope is the proportion of one particular isotope in a mixture of isotopes found in nature
- For example, the relative abundance of Cl-35 and Cl-37 is 75% and 25% respectively
- This means that in nature, 75% of the chlorine atoms is the Cl-35 isotope and 25% is the Cl-37 isotope
- The heights of the peaks in mass spectroscopy show the proportion of each isotope present
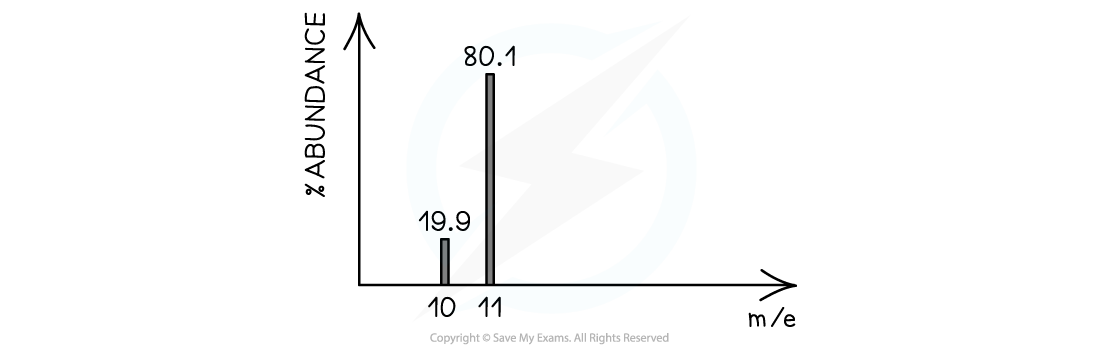
The peak heights show the relative abundance of the boron isotopes: boron-10 has a relative abundance of 19.9% and boron-11 has a relative abundance of 80.1%
Worked example: Calculating m/e ratio

Answer
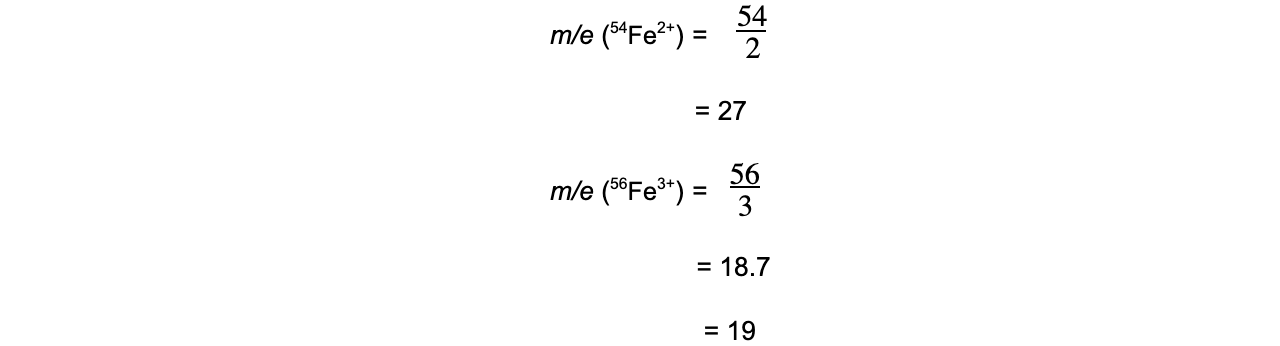
56Fe3+ has a smaller m/e ratio and will therefore be deflected more.
It also has the largest positive charge and will be more attracted to the negative pole of the magnet within the mass spectrometer.
Exam Tip
A small m/e value corresponds to fragments that are either small or have a high positive charge or a combination of both.
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





