- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
CIE A Level Biology复习笔记2.2.1 Biological Molecules: Key Terms
Biological Molecules: Key Terms
- The key molecules that are required to build structures that enable organisms to function are:
- Carbohydrates
- Proteins
- Lipids
- Nucleic Acids
- Water
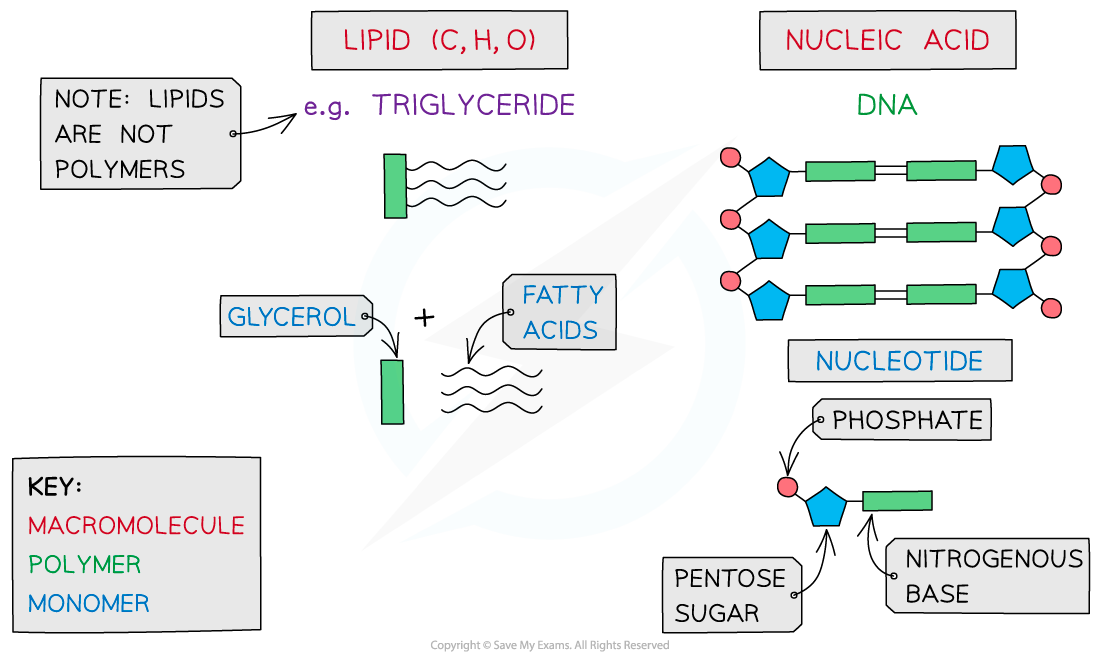
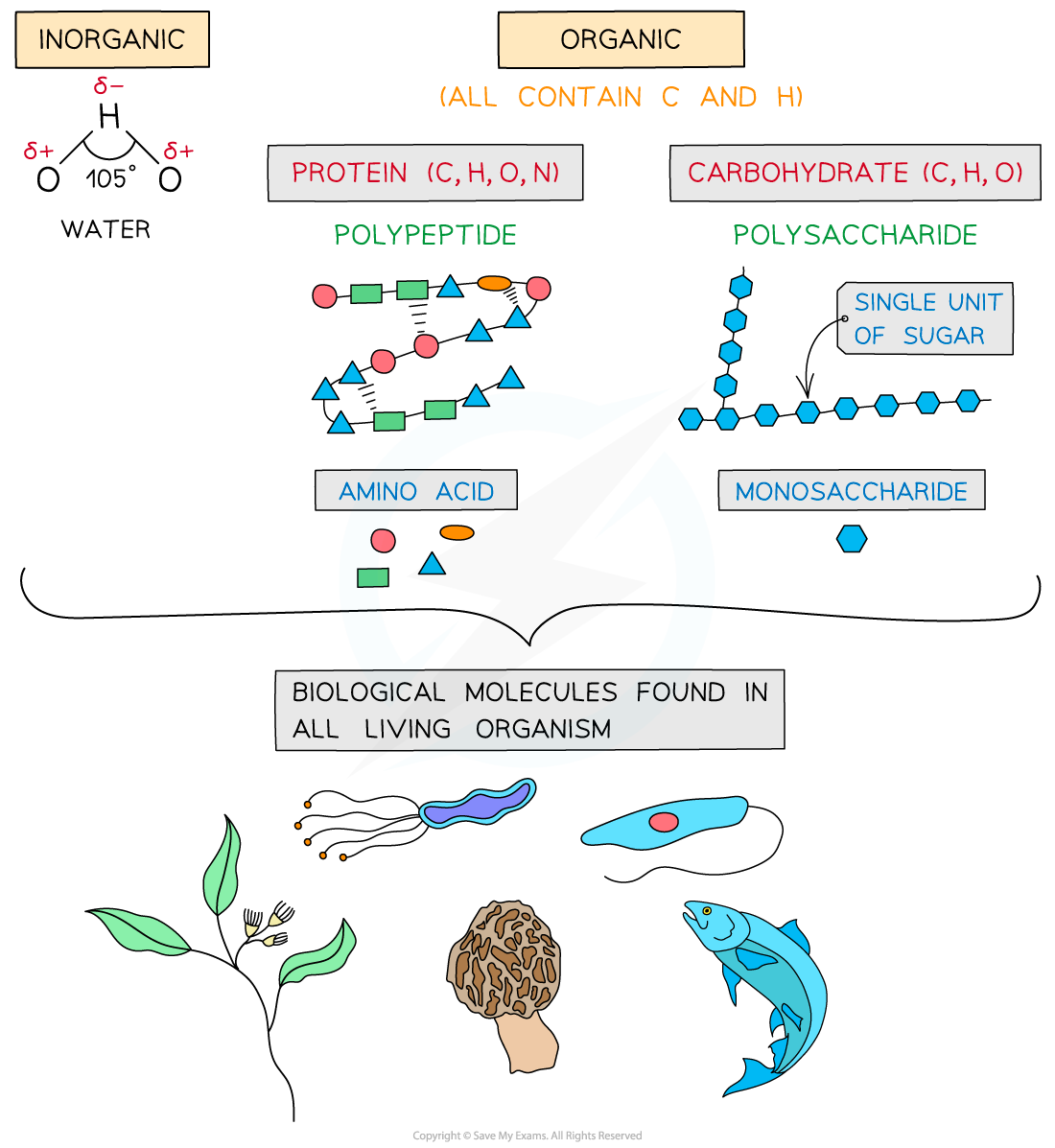
The key biological molecules for living organisms
- Carbohydrates, proteins, lipids and nucleic acids contain the elements carbon (C) and hydrogen (H) making them organic compounds
- Carbon atoms are key to the organic compounds because:
- Each carbon atom can form four covalent bonds – this makes the compounds very stable (as covalent bonds are so strong they require a large input of energy to break them)
- Carbon atoms can form covalent bonds with oxygen, nitrogen and sulfur
- Carbon atoms can bond to form straight chains, branched chains or rings
- Carbon compounds can form small single subunits (monomers) that bond with many repeating subunits to form large molecules (polymers) by a process called polymerisation
- Macromolecules are very large molecules
- That contain 1000 or more atoms therefore having a high molecular mass
- Polymers can be macromolecules, however not all macromolecules are polymers as the subunits of polymers have to be the same repeating units
Carbohydrates
- Carbohydrates are one of the main carbon-based compounds in living organisms
- All molecules in this group contain C, H and O
- As H and O atoms are always present in the ratio of 2:1 (eg. water H2O, which is where ‘hydrate’ comes from) they can be represented by the formula Cx (H2O)y
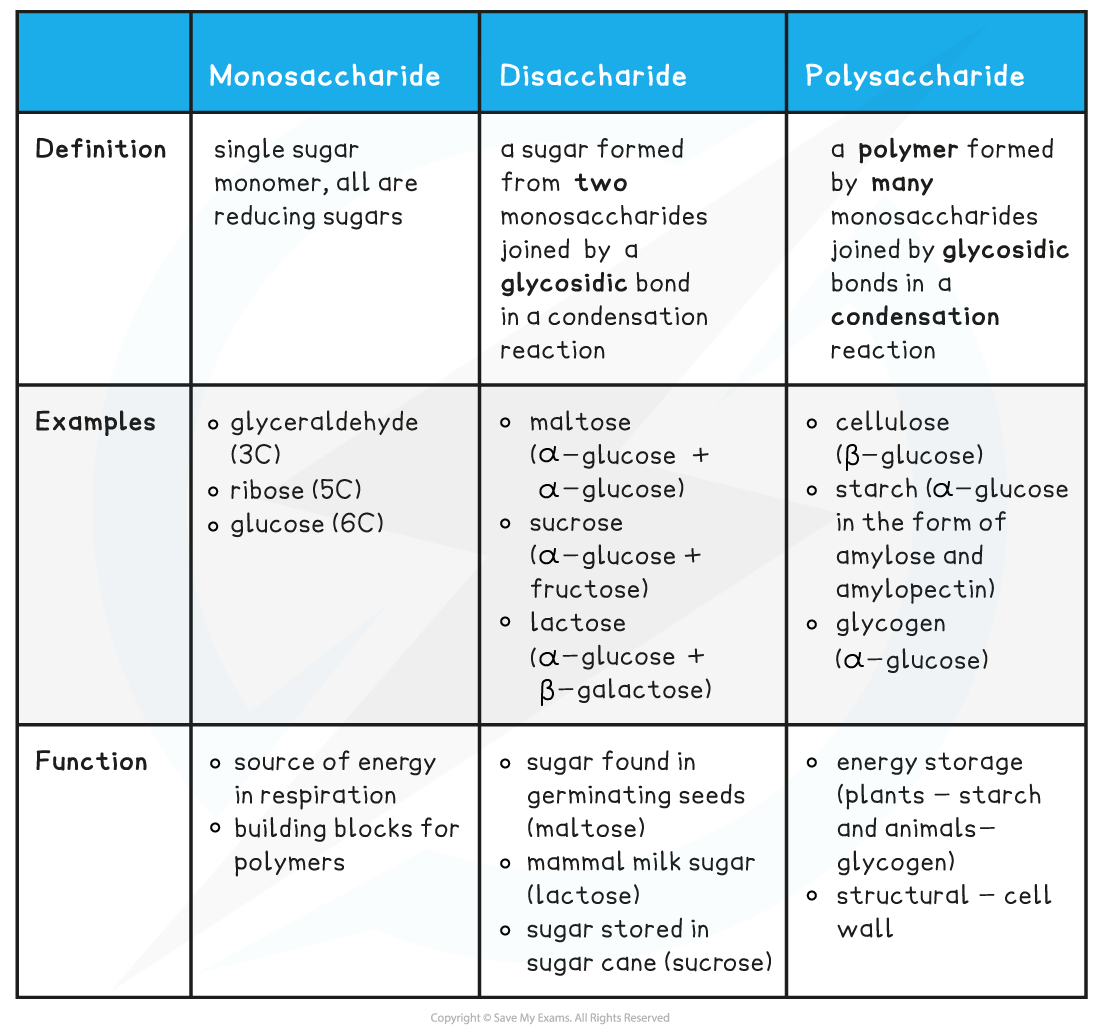
- The three types of carbohydrates are monosaccharides, disaccharides and polysaccharides
Types of carbohydrate table
Exam Tip
When discussing monomers and polymers, give the definition but also name specific examples eg. nucleic acids – the monomer is a nucleotide.
The Two Forms of Glucose
- The most well-known carbohydrate monomer is glucose
- Glucose has the molecular formula C6H12O6
- Glucose is the most common monosaccharide and is of central importance to most forms of life
- There are different types of monosaccharide formed from molecules with varying numbers of carbon atom, for example:
- Trioses (3C) eg. glyceraldehyde
- Pentoses (5C) eg. ribose
- Hexoses (6C) eg. glucose
- Glucose exists in two structurally different forms – alpha (α) glucose and beta (β) glucose and is therefore known as an isomer
- This structural variety results in different functions between carbohydrates
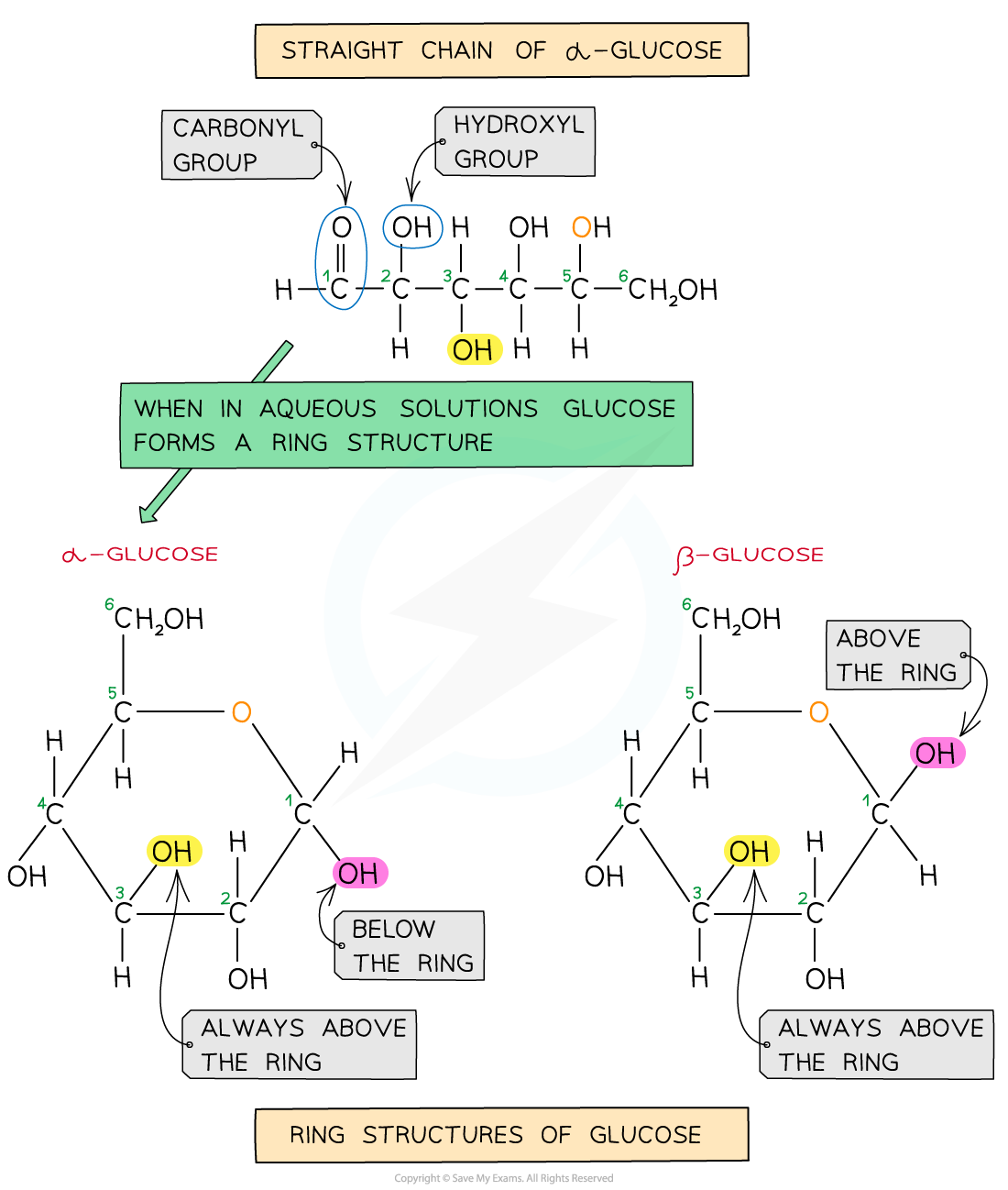
Straight chain and ring structural formula of alpha & beta glucose
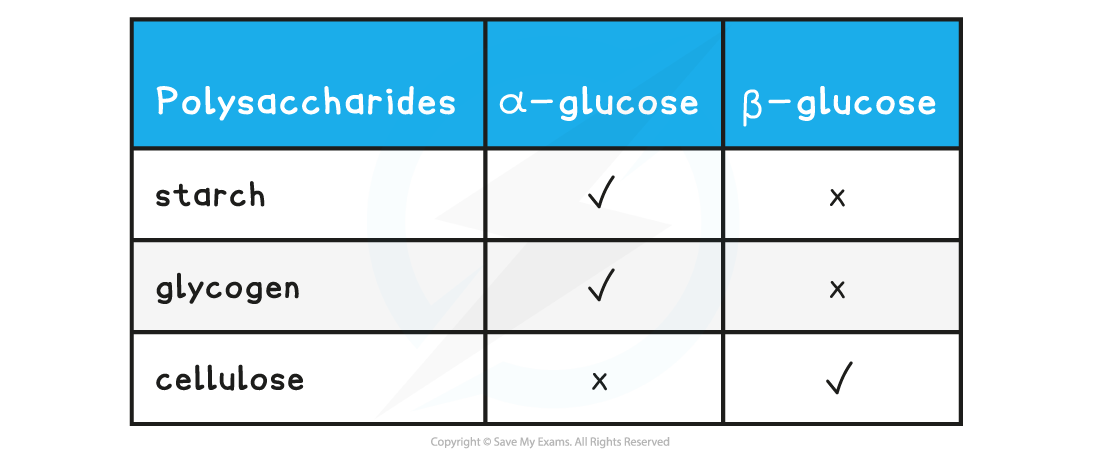
- Different polysaccharides are formed from the two isomers of glucose
Structure of polysaccharides table
Exam Tip
You must be able to recognise and draw the isomers of α and β glucose.
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





