- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
IB DP Chemistry: SL复习笔记9.1.1 Oxidation & reduction
Oxidation & Reduction
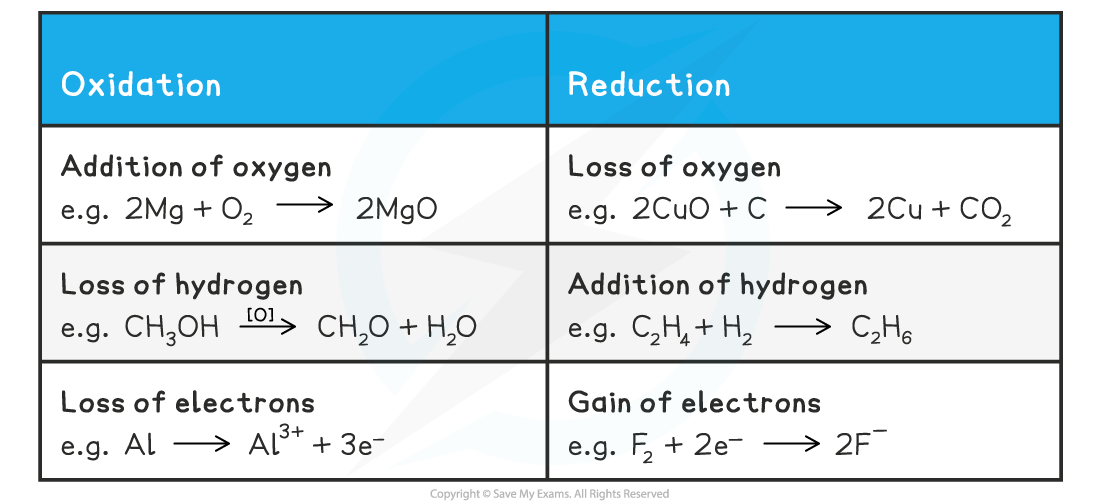
- There are three definitions of oxidation and reduction used in different branches of chemistry
- Oxidation and reduction can be used to describe any of the following processes
Definitions and Examples of Oxidation & Reduction Table
Oxidation Numbers
- The oxidation number of an atom is the charge that would exist on an individual atom if the bonding were completely ionic
- It is like the electronic ‘status’ of an element
- Oxidation numbers are used to...
-
- tell if oxidation or reduction has taken place
- work out what has been oxidised and/or reduced
- construct half equations and balance redox equations
Atoms and simple ions
- The oxidation number is the number of electrons which must be added or removed to become neutral
- The oxidation number is always written with the charge before the number
Oxidation Number of Simple Ions Table

Worked Example
What are the oxidation states of the elements in the following species?
a) C b) Fe3+ c) Fe2+ d) O2- e) He f) Al3+
Answers:
a) 0 b) +3 c) +2 d) -2 e) 0 f) +3
- So, in simple ions, the oxidation number of the atom is the charge on the ion:
- Na+, K+, H+ all have an oxidation number of +1
- Mg2+, Ca2+, Pb2+ all have an oxidation number of +2
- Cl-, Br-, I- all have an oxidation number of -1
- O2-, S2- all have an oxidation number of -2
Molecules or Compounds
- In molecules or compounds, the sum of the oxidation numbers on the atoms is zero
Oxidation Number in Molecules or Compounds Table

- Because CO2 is a neutral molecule, the sum of the oxidation states must be zero
- For this, one element must have a positive oxidation number and the other must be negative
How do you determine which is the positive one?
- the more electronegative species will have the negative value
- electronegativity increases across a period and decreases down a group
- O is further to the right than C in the periodic table so it has the negative value
How do you determine the value of an element's oxidation state?
- from its position in the periodic table and/or
- the other element(s) present in the formula
Variable Oxidation Numbers
- Many atoms, such as S, N and Cl, can exist in a variety of oxidation states
- The oxidation number of these atoms can be calculated by assuming that the oxidation number of the other atom is fixed
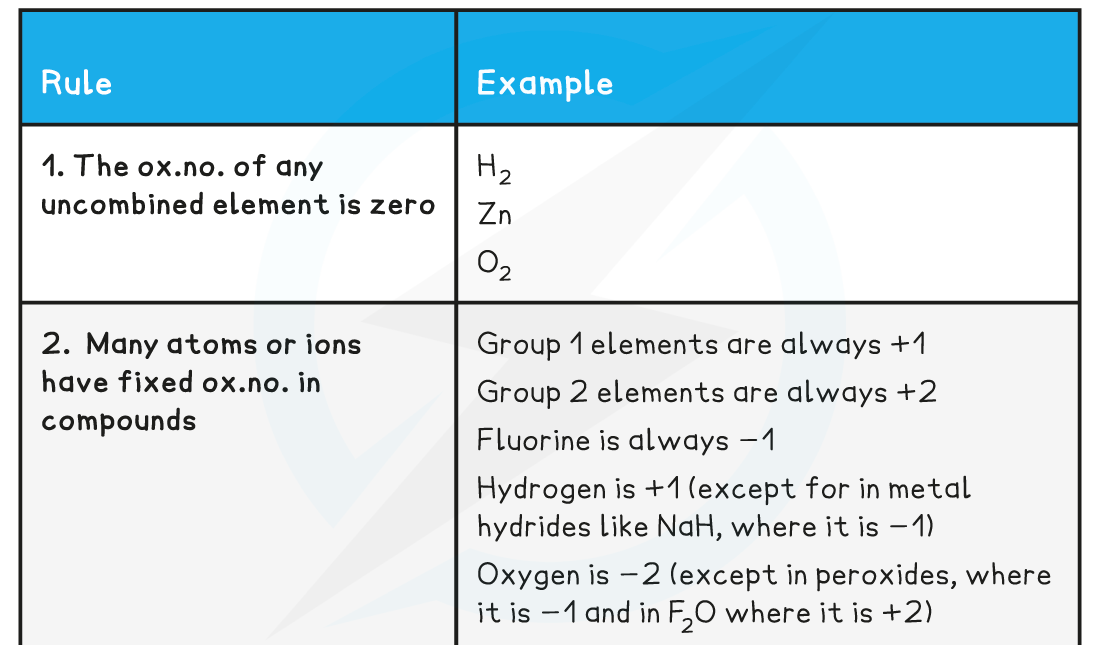
- Here are six rules to deduce the oxidation number of an element
Oxidation Number Rules Table

Exam Tip
Oxidation number and oxidation state are often used interchangeably, though IUPAC does not distinguish between the two terms.Oxidation numbers are represented by Roman numerals according to IUPAC
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





