- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
IB DP Chemistry: SL复习笔记8.3.2 Effects of Acid Deposition
Effects of acid deposition
Effect on materials
- Acid deposition can react with metals and rocks (such as limestone) causing buildings and statues to get damaged
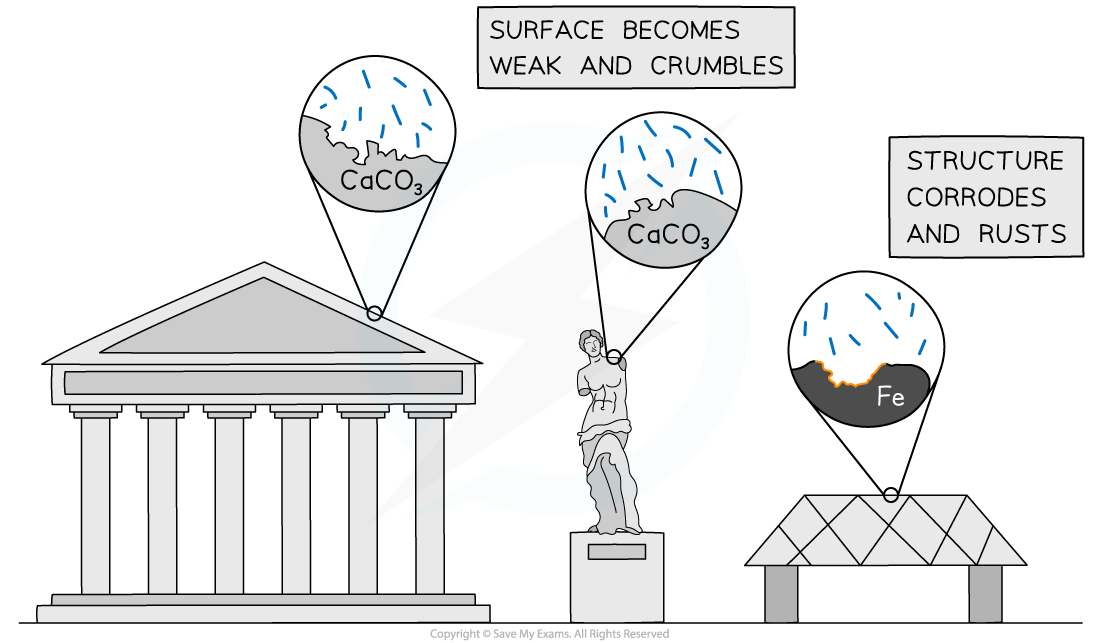 The impact of acid deposition can be seen on buildings, statues and metallic structures, particularly in polluted cities
The impact of acid deposition can be seen on buildings, statues and metallic structures, particularly in polluted cities
- Limestone and marble both contain calcium carbonate
- The calcium carbonate reacts with sulfuric or nitric acids causing stonework to corrode and weaken:
CaCO3 (s) + H2SO4 (aq) → CaSO4 (aq) + CO2 (g) + H2O (l)
CaCO3 (s) + 2HNO3 (aq) → Ca(NO3)2(aq) + CO2 (g) + H2O (l)
- In addition dry deposition also affects limestone:
CaCO3 (s) + H2SO4 (aq) → CaSO4 (aq) + CO2 (g) + H2O (l)
- Metallic structures, such as iron bridges, are vulnerable to corrosion by wet and dry deposition forming the salt of the metal:
Fe (s) + H2SO4 (aq) → FeSO4 (aq) + H2 (g)
Fe (s) + SO2 (g) + O2 (g) → FeSO4 (s)
Effect on plants
- Apart from acid deposition directly falling on leaves and killing plants, acid particulates can block stomata ( plant pores) and prevent gaseous exchange
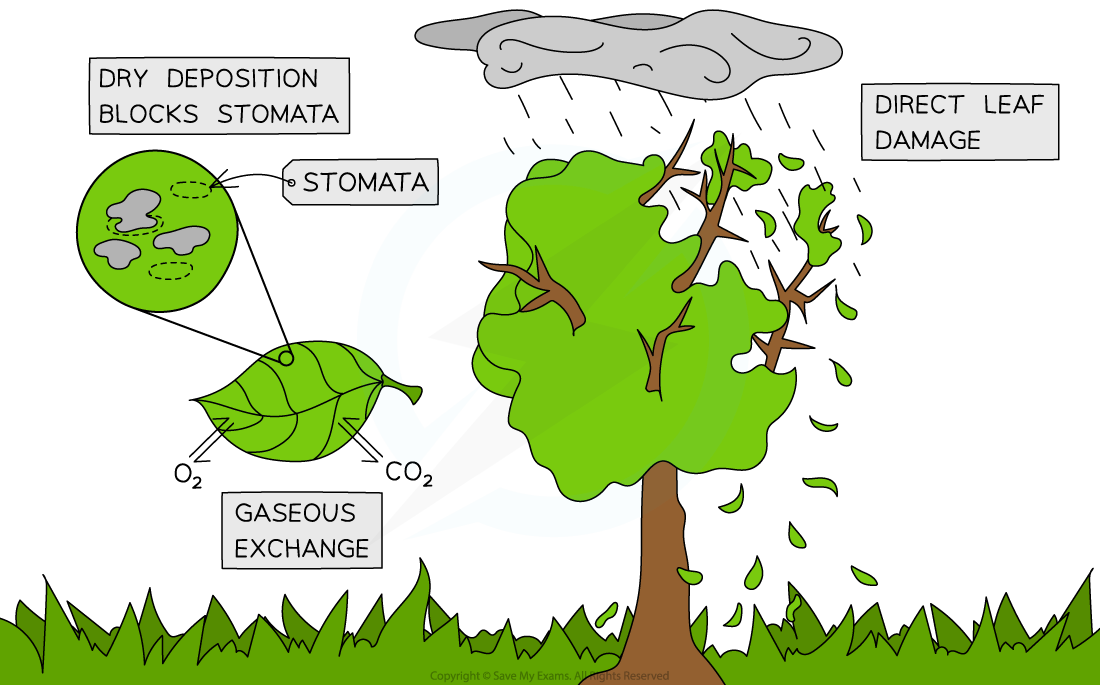 The impact of acid deposition on plant life
The impact of acid deposition on plant life
- Acid rain can fall on soils and release important minerals such as magnesium, calcium and potassium which are leached (washed out) from soils and are therefore unavailable to plants
- Aluminium ions released from rocks are toxic to many plants and damage their roots
Effect on water
- When acid rain falls on rivers and lakes the pH can fall to levels that are unable to support life

The impact of acid deposition on waterways
- Below about pH 4, aluminium ions are released from rocks when they are held as aluminium hydroxide
Al(OH)3 (s) + 3H+ (aq) → Al3+ (aq) + 3H2O (l)
- Alumnium ions are toxic to fish as they damage the gills and prevent fish from efficiently absorbing oxygen
- Nitrate ions from nitric acid in acid rain can contibute to over-fertilization of waterways and lead to eutrophication
- Eutrophication is excessive algal growth that results in oxygen depletion and stagnation of waterways
Effect on human health
- Although acid rain is too dilute to cause any direct impact on the skin, acidic particulates in the air can increase the risk of respiratory diseases such as bronchitis, asthma and emphysema
- When acid rain comes into contact with metal pipes there is an increased risk that toxic metal ions will be released into the water supply such as Cu2+, Al3+ and Pb2+
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





