- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
IB DP Chemistry: SL复习笔记2.1.4 The Electromagnetic Spectrum
The Electromagnetic Spectrum
- The electromagnetic spectrum is a range of frequencies that covers all electromagnetic radiation and their respective wavelengths and energy
- It is divided into bands or regions, and is very important in analytical chemistry
- The spectrum shows the relationship between frequency, wavelength and energy
- Frequency is how many waves pass per second, and wavelength is the distance between two consecutive peaks on the wave
- Gamma rays, X-rays and UV radiation are all dangerous - you can see from that end of the spectrum that it is high frequency and high energy, which can be very damaging to your health
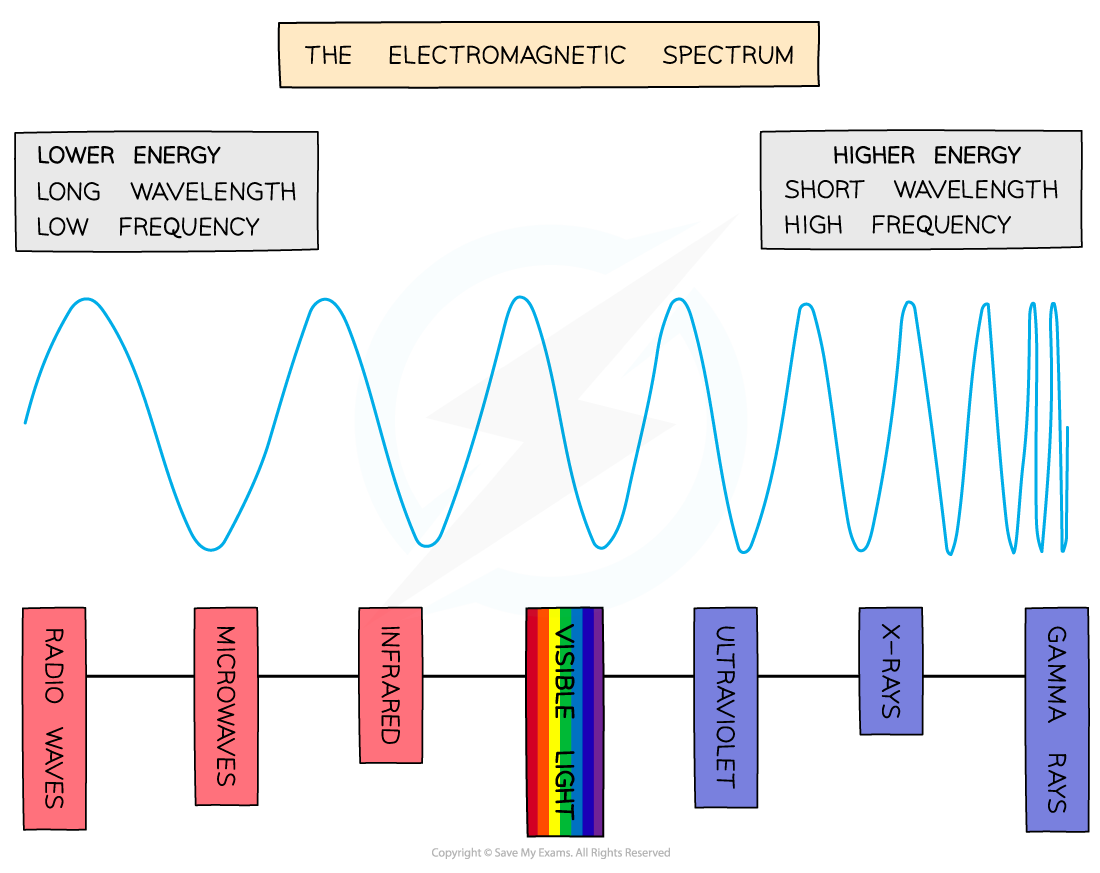
- All light waves travel at the same speed; what distinguishes them is their different frequencies
- The speed of light (symbol ‘c’) is constant and has a value of 3.00 x 108 ms-1
- As you can see from the spectrum, frequency (symbol ‘ν') is inversely proportional to wavelength (symbol ‘λ')
- In other words, the higher the frequency, the shorter the wavelength
- The equation that links them is c = νλ
- Since c is constant you can use the formula to calculate the frequency of radiation given the wavelength, and vice versa
Continuous versus line spectrum
- A continuous spectrum in the visible region contains all the colours of the spectrum
- This is what you are seeing in a rainbow, which is formed by the refraction of white light through a prism or water droplets in rain

A continuous spectrum shows all frequencies of light
- However, a line spectrum only shows certain frequencies

The line spectrum of helium which shows only certain frequencies of light
- This tells us that the emitted light from atoms can only be certain fixed frequencies - it is quantised (quanta means 'little packet')
- Electrons can only possess certain amounts of energy - they cannot have any energy value
Exam Tip
The formula that relates frequency and wavelength is printed in Section 1 of the IB Chemistry Data Booklet so you don’t need to learn itYou will also find the speed of light and other useful constants in Section 2
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





