- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
IB DP Chemistry: SL复习笔记1.1.4 The Mole Concept
The Mole
- The Avogadro constant (NA or L) is the number of particles equivalent to the relative atomic mass or molecular mass of a substance in grams
- The Avogadro constant applies to atoms, molecules and ions
- The value of the Avogadro constant is 6.02 x 1023 g mol-1
- The mass of a substance with this number of particles is called the molar mass
- One mole of a substance contains the same number of fundamental units as there are atoms in exactly 12.00 g of 12C
- If you had 6.02 x 1023 atoms of carbon-12 in your hand, you would have a mass of exactly 12.00 g
- One mole of water would have a mass of (2 x 1.01 + 16.00) = 18.02 g
Worked Example
Determine the number of atoms, molecules and the relative mass of 1 mole of:
- Na
- H2
- NaCl
Answer 1:
-
- The relative atomic mass of Na is 22.99
- Therefore, 1 mol of Na has a mass of 22.99 g mol-1
- 1 mol of Na will contain 6.02 x 1023 atoms of Na (Avogadro’s constant)
Answer 2:
-
- The relative atomic mass of H is 1.01
- Since there are 2 H atoms in H2, the mass of 1 mol of H2 is (2 x 1.01) 2.02 g mol-1
- 1 mol of H2 will contain 6.02 x 1023 molecules of H2
- However, since there are 2 H atoms in each molecule of H2, 1 mol of H2 molecules will contain 1.204 x 1024 H atoms
Answer 3:
-
- The relative atomic masses of Na and Cl are 22.99 and 35.45 respectively
- Therefore, 1 mol of NaCl has a mass of (22.99 + 35.45) 58.44 g mol-1
- 1 mol of NaCl will contain 6.02 x 1023 formula units of NaCl
- Since there is both an Na and a Cl atom in NaCl, 1 mol of NaCl will contain 1.204 x 1024 atoms in total

Relative Mass
Relative atomic mass, Ar
- The relative atomic mass (Ar) of an element is the weighted average mass of one atom compared to one twelfth the mass of a carbon-12 atom
- The relative atomic mass is determined by using the weighted average mass of the isotopes of a particular element
- The Ar has no units as it is a ratio and the units cancel each other out
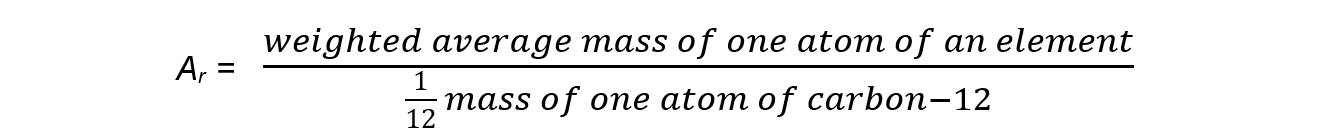
Relative isotopic mass
- The relative isotopic mass is the mass of a particular atom of an isotope compared to one twelfth the mass of a carbon-12 atom
- Atoms of the same element with a different number of neutrons are called isotopes
- Isotopes are represented by writing the mass number as 20Ne, or neon-20 or Ne-20
- To calculate the average atomic mass of an element the percentage abundance is taken into account
- Multiply the atomic mass by the percentage abundance for each isotope and add them all together
- Divide by 100 to get average relative atomic mass
- This is known as the weighted average of the masses of the isotopes
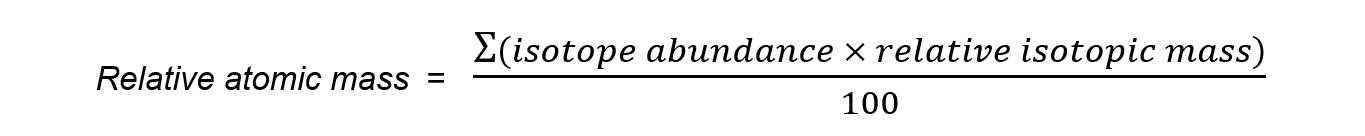
Relative molecular mass, Mr
- The relative molecular mass (Mr) is the weighted average mass of a molecule compared to one twelfth the mass of a carbon-12 atom
- The Mr has no units
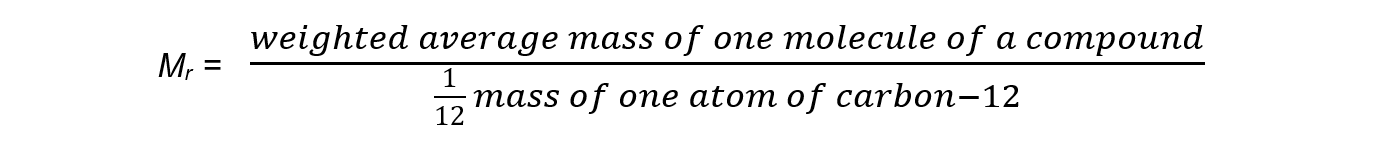
- The Mr can be found by adding up the relative atomic masses of all atoms present in one molecule
- When calculating the Mr the simplest formula for the compound is used, also known as the formula unit
- E.g. Silicon dioxide has a giant covalent structure, but the simplest formula (the formula unit) is SiO2
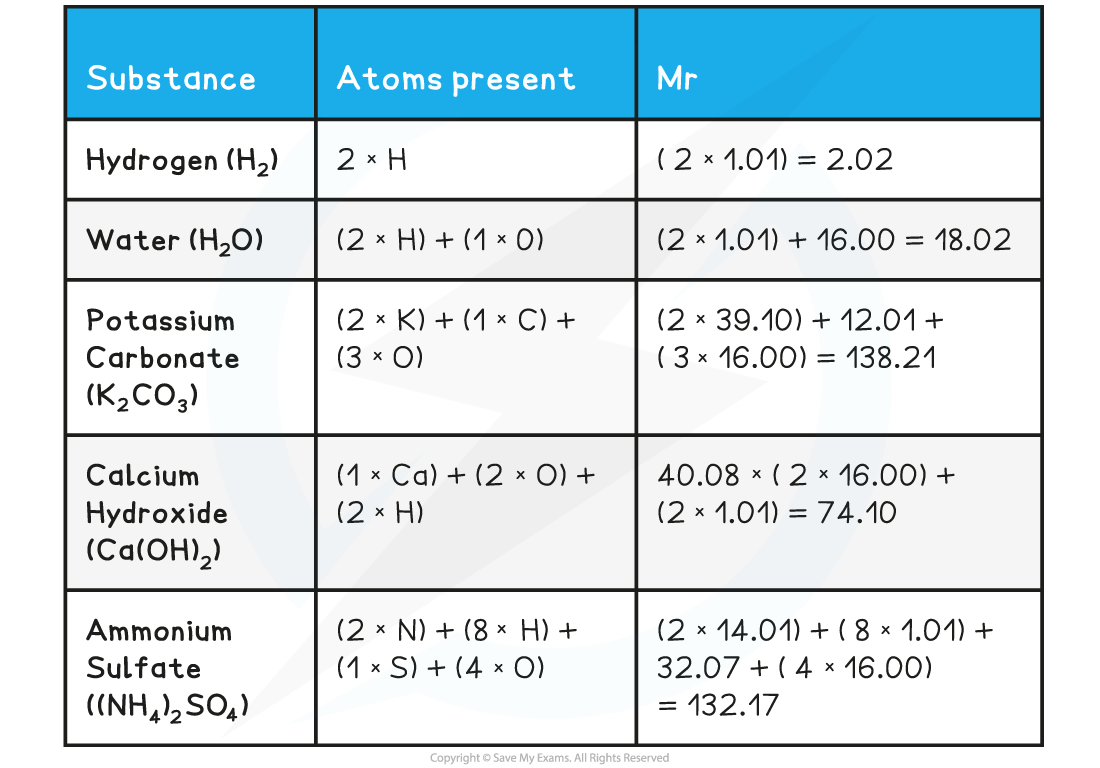
Relative formula mass, Mr
- The relative formula mass (Mr) is used for compounds containing ions
- It has the same units and is calculated in the same way as the relative molecular mass
- In the table above, the Mr for potassium carbonate, calcium hydroxide and ammonium sulfate are relative formula masses
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





