- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
IB DP Biology: SL复习笔记4.2.4 Carbon Cycle: Organic Matter
Formation of Peat
- Under normal conditions the tissues of dead organisms are completely broken down by decomposers known as saprotrophs and detritivores
- These decomposers require specific conditions, such as oxygen availability and the correct pH
- If ground is waterlogged or acidic, most decomposers cannot survive, and only partial decomposition takes place
- Waterlogging occurs when levels of rainfall exceed levels of evaporation
- When ground is waterlogged, the air spaces in the soil is filled with water, and the conditions become anaerobic
- Low pH can occur due to mineral leaching, acidic bedrock, bacterial activity, and the growth of certain types of plant
- Waterlogging occurs when levels of rainfall exceed levels of evaporation
- Under waterlogged and acidic conditions partly decomposed organic material, such as dead plant matter, accumulates, and becomes compacted under its own weight over time; this compacted, partially decomposed plant matter forms peat
- The place where peat accumulates is known as a peat bog, or peatland
- There are various types of peat bog e.g. blanket bog and raised bog, depending on the landscape and conditions in which it forms
- The carbon compounds in plant material are trapped in peat
- Over millions of years, peat can develop into the fossil fuels coal, oil, or natural gas
- Peat is considered to be a valuable resource, and digging up peat bogs has been a common practice
- Peat can be burned as a fuel due to its carbon content
- The high nutrient content of peat means that it is often used in compost
- The importance of peat bogs as a carbon store and unique, biodiverse, habitat is now better understood, and many people work to protect and restore peat bogs
Exam Tip
Make sure you know that peat is formed from partly decomposed plant matter that results from acidic and waterlogged (anaerobic) conditions.
Fossilisation
- If animals and plants die in acidic or anaerobic conditions then complete decomposition cannot take place
- Micro-organisms that carry out decomposition cannot survive in acidic or anaerobic conditions
- The carbon in remaining tissues can be converted into fossil fuels over millions of years
- This means that carbon taken into the tissues of living organisms during past geological eras is locked up in fossil fuels
- Coal forms when peat (see above) that forms in acidic and anaerobic conditions is buried, compressed, and heated e.g. after a rise in sea levels
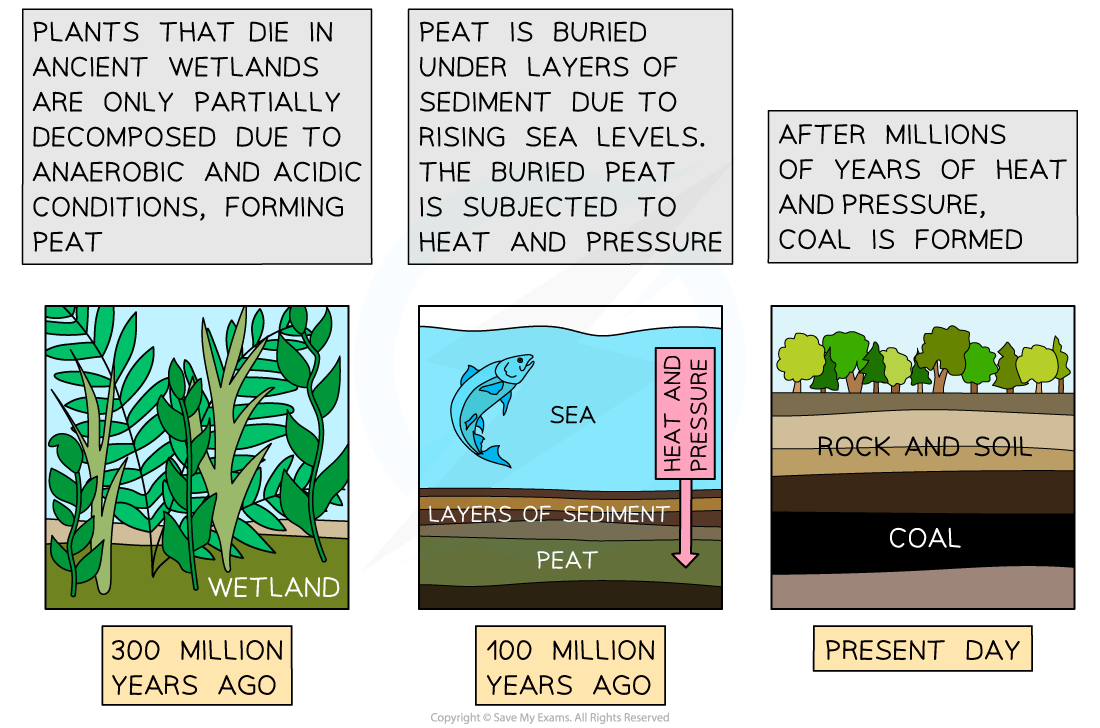
Formation of coal from peat
- Oil and natural gas form in anaerobic conditions at the bottom of seas and lakes when aquatic organisms die and are covered by layers of sediment, and are then compressed and heated.
- Natural gas forms deposits in porous rock when surrounding non-porous rock prevents it from escaping to the surface
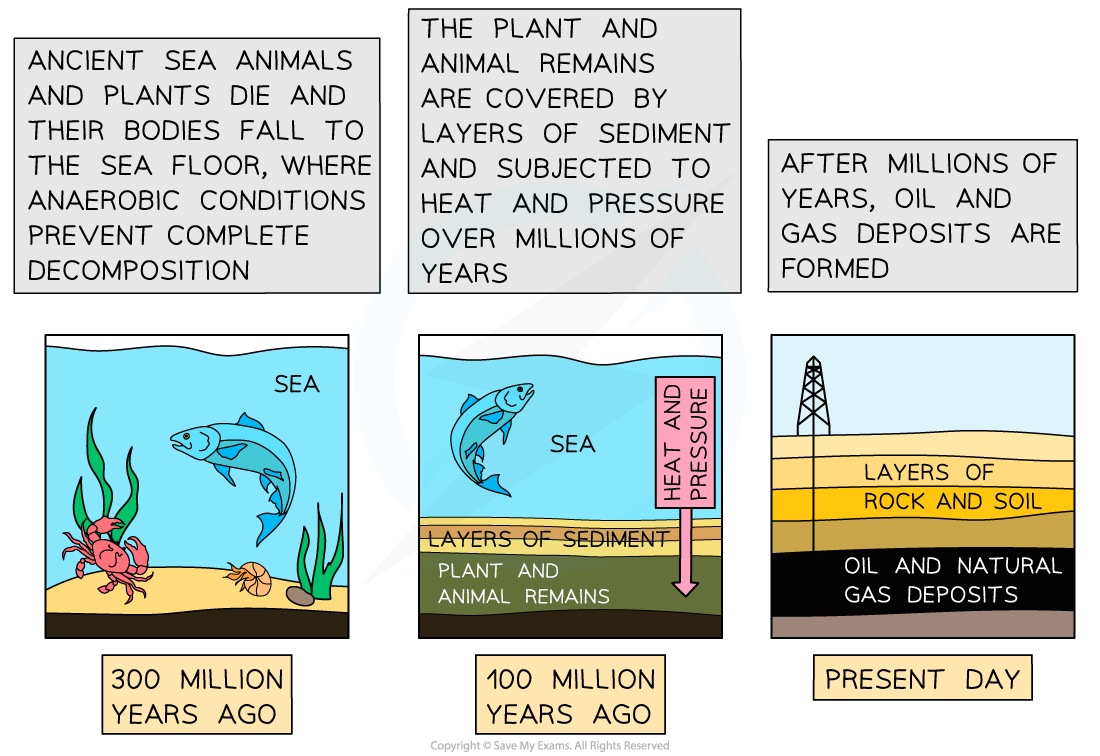
Formation of oil and gas from the bodies of aquatic organisms
- When fossil fuels are burned (the process is known as combustion), the carbon combines with oxygen and carbon dioxide is released into the atmosphere
- Increased use of fossil fuels is contributing to an increase in the carbon dioxide content of the atmosphere
Limestone Formation
- Many marine (sea-living) organisms extract carbon from their surroundings in order to produce calcium carbonate, from which they build certain body parts. For example:
- The shells of molluscs (e.g. mussels and clams)
- The hard bodies (exoskeletons) of corals
- When these organisms die, the soft parts of their bodies are broken down by decomposers, but their calcium carbonate outer shells remain and fall to the seafloor
- The layers of sediment that form on the seafloor are rich in the calcium carbonate shells of marine organisms, and as the layers are compacted with their own weight and that of the sea above them, they harden, and limestone is formed
- The carbon in the hard shells of these marine organisms is locked up in limestone rocks e.g. chalk
- Ocean acidification can cause the calcium carbonate shells of marine organisms to dissolve, releasing this carbon back into the surrounding water
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





