- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
IB DP Biology: SL复习笔记2.3.4 Skills: Molecules
Molecular Diagrams: Drawing
Drawing biological molecules
- It is important to be able to draw a few key molecules
- There is a huge variety of biological molecules, but only the most important ones are required
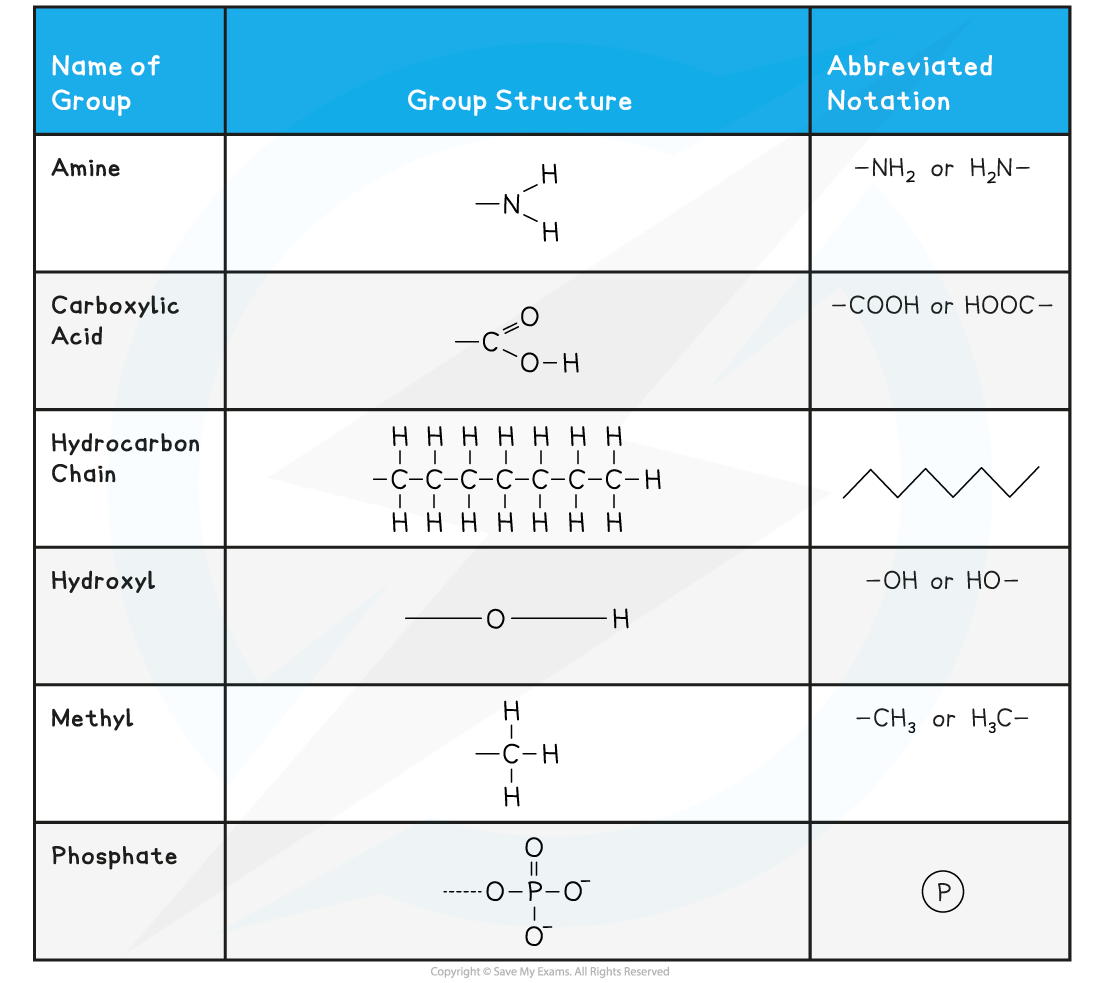
- Element symbols from the Periodic Table are used
- A short, straight line is used for a covalent bond, with two lines for a double bond
- Some chemical groups may be denoted by a symbol such as ℗ for a phosphate group
- The symbol R represents a variable chemical group, such as the variable side groups of amino acids
- An exam question may require you to draw various molecules
- It's advisable to break the task down into stages
Symbols Used in Biological Molecule Drawings Table
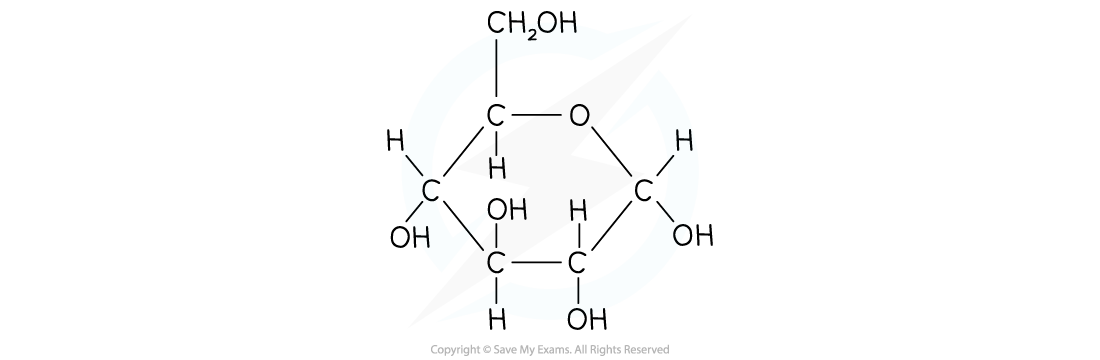
Drawing α-D-glucose
- Aspects to remember
- Glucose has the formula C6H12O6
- In solid form, glucose has a linear structure
- It forms a hexagonal ring in an aqueous solution
- As aqueous glucose is the only state that glucose exists in biology, it's the ring structure that should be learned
- One of the corners of the ring (draw this in the top-right) is occupied by an oxygen atom
- The 6th carbon occupies a side chain (top-left)
- The carbon atoms are numbered 1 to 6 starting on the right and working clockwise
- The hydroxyl groups occupy positions above or below the ring as follows
- Carbon atom 1 - below
- Carbon atom 2 - below
- Carbon atom 3 - above
- Carbon atom 4 - below
- You can ignore the 'D' in the names alpha-D-glucose or beta-D-glucose
- The only other version is L-glucose which plays no significant role in biology
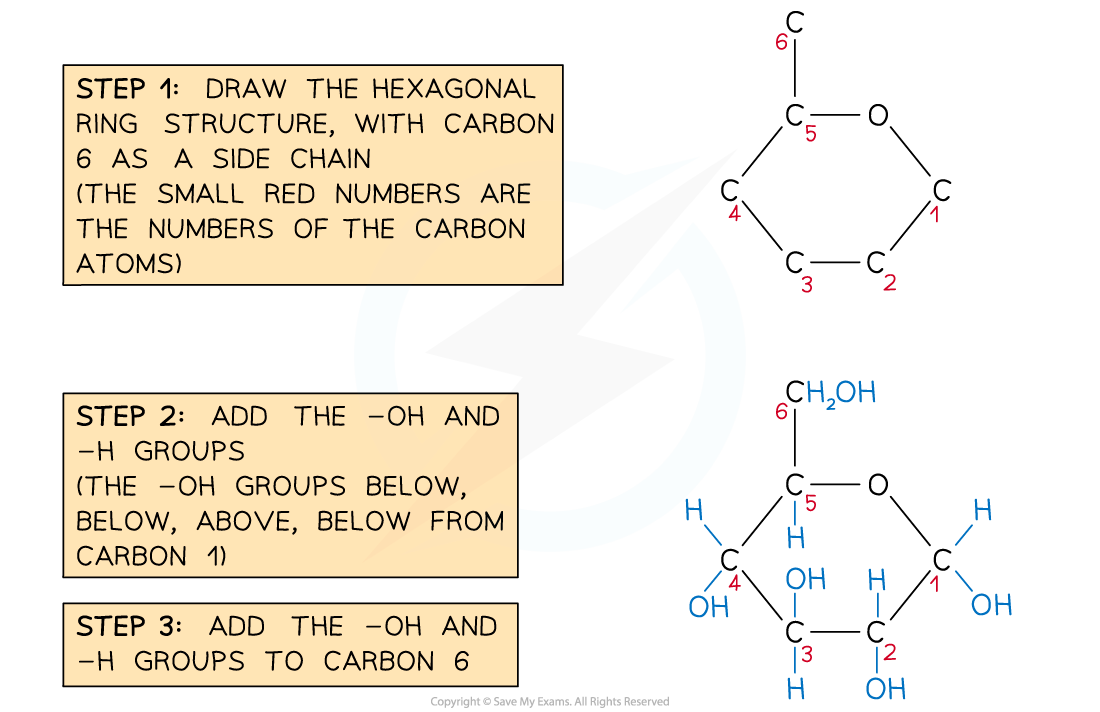
Recommended steps to draw a molecule of α-D-glucose
Structure of α-D-glucose
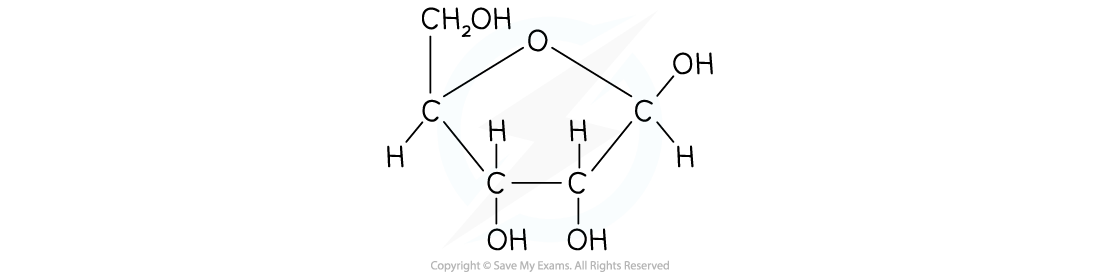
β-D-glucose is very slightly different in structure
- Beta-glucose (β-glucose) has a small, subtle difference to α-glucose
- The hydroxyl group on carbon atom 1 sits ABOVE the ring, rather than below
- This sugar is the monomer of cellulose
- This example of two different isomers changes the properties of the polysaccharide formed from these monomers drastically
- It accounts for all the many differences between starch and cellulose
- The hydroxyl groups occupy positions above or below the ring as follows
- Carbon atom 1 - above
- Carbon atom 2 - below
- Carbon atom 3 - above
- Carbon atom 4 - below
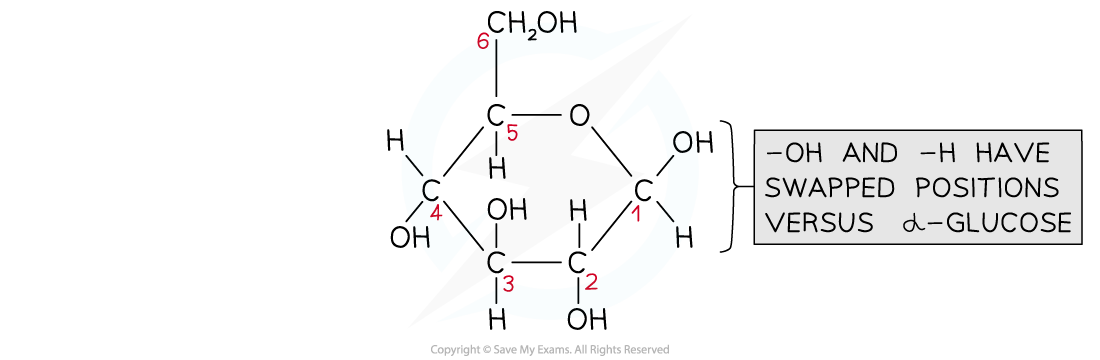
Structure of β-D-glucose
Drawing a ribose sugar
- This family of sugars play a role in DNA and RNA structure, as well as ATP
- Ribose is a form of pentose sugar (5 carbon atoms)
- Like glucose, ribose has a ring structure
- Aspects to remember
- Ribose has the formula C5H10O5
- It forms a pentagonal ring in an aqueous solution
- One of the corners of the ring (draw this in the top) is occupied by an oxygen atom
- The 5th carbon occupies a side chain (top-left)
- The carbon atoms are numbered 1 to 5 starting on the right and working clockwise
- The hydroxyl groups occupy positions above or below the ring as follows
- Carbon atom 1 - above
- Carbon atom 2 - below
- Carbon atom 3 - below
- Ribose sugars have an important close relative - deoxyribose sugars
- Both are key components of RNA and DNA respectively
- The 'R' and 'D' of RNA and DNA comes from the sugar in the structure, ribose or deoxyribose
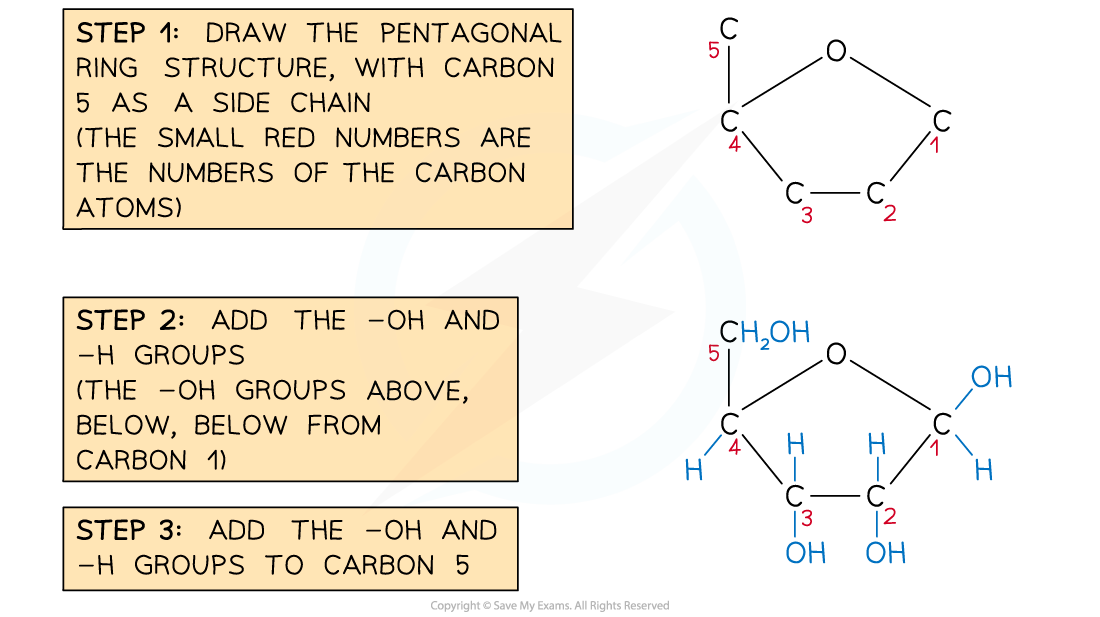
Recommended steps to draw a molecule of ribose
Structure of ribose
Drawing a saturated fatty acid
- There are two aspects to a saturated fatty acid
- A saturated hydrocarbon chain
- Contains only C-C single bonds
- Each internal carbon atom is bonded to 2 hydrogen atoms
- A carboxylic acid group at one end
- A saturated hydrocarbon chain
- You don't need to memorise any names of saturated fatty acids
- The number of carbon atoms in your chain is also not important, but greater than around 8 is advised.
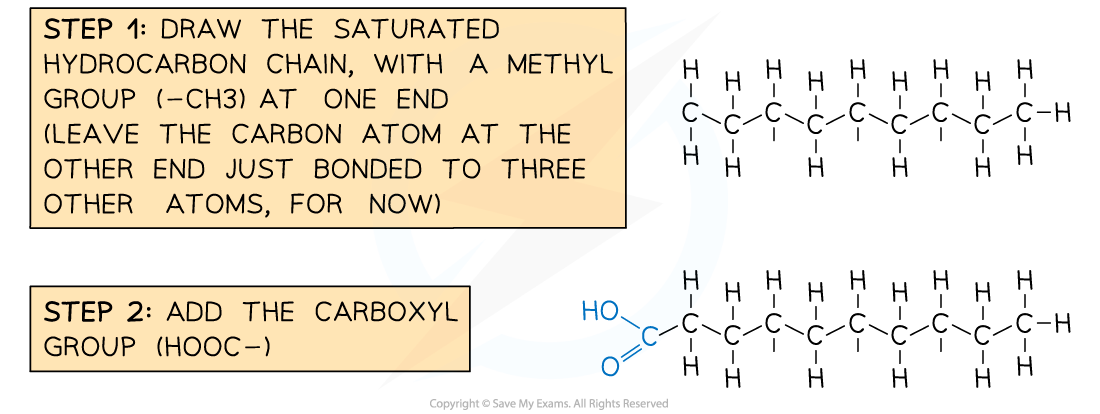
Recommended steps to draw a molecule of a saturated fatty acid
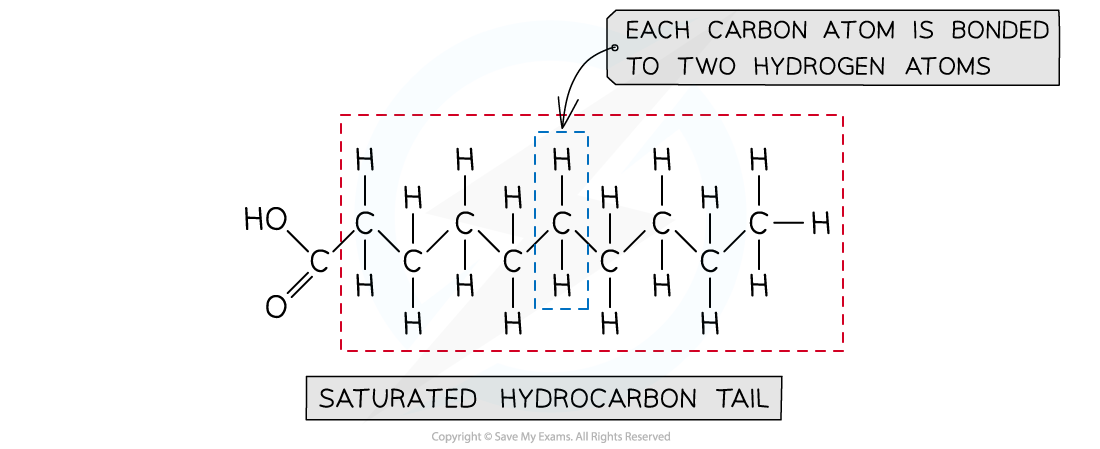
A saturated fatty acid
Drawing a generalised amino acid
- Each amino acid has central carbon atom
- Three of the bonds from the central carbon atom are occupied as follows
- a hydrogen atom
- a carboxylic acid group
- an amine group
- The fourth bond attaches the central carbon to the R group
- The R group is variable and determines the identity of the amino acid
- You won't need to remember any of the R groups or amino acid names
- Drawing the 4 groups surrounding the central carbon in a flat structure is acceptable, although the real arrangement of bonds around a carbon atom is in a tetrahedral shape
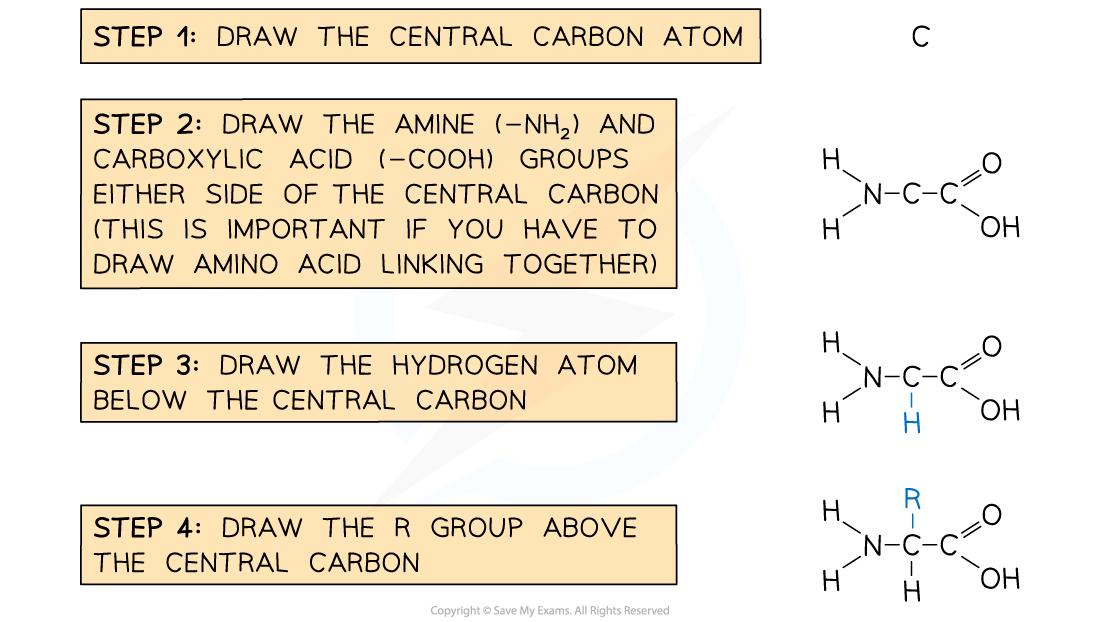
Recommended steps to draw a molecule of generalised amino acid
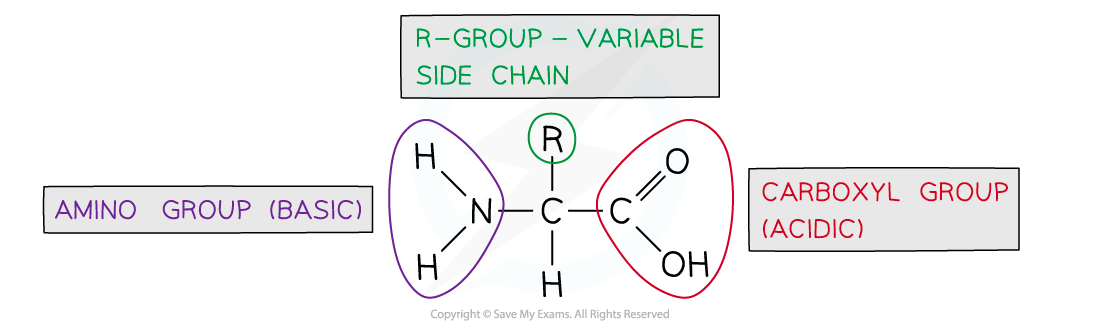
The generalised structure of an amino acid
Exam Tip
The specification is very clear about what you should remember and equally importantly, what you don't have to memorise in this topic.With all these drawings, practise with a pencil and plenty of paper until you get it right. It WILL stick! Once you've memorised it, 'draw' questions should be easy marks!
Molecular Diagrams: Identification
- As well as being able to draw certain molecules, an important skill is being able to recognise certain biochemicals from molecular diagrams
- There are several features that help to identify molecules
- The presence of carbon, hydrogen, oxygen, nitrogen, sulfur and phosphorus can help in the identification
- All biological macromolecules contain carbon, hydrogen and oxygen
- Nitrogen and sulfur are present in proteins
- Nitrogen is present in nucleic acids (DNA, RNA)
- Phosphorus is also present in certain molecules (DNA, RNA and phospholipids)
- The presence of ring structures, hydrocarbon chains, carbon-to-carbon double bonds, double-stranded areas and the ratio of carbon to oxygen in a molecule all give clues about the molecule's identity
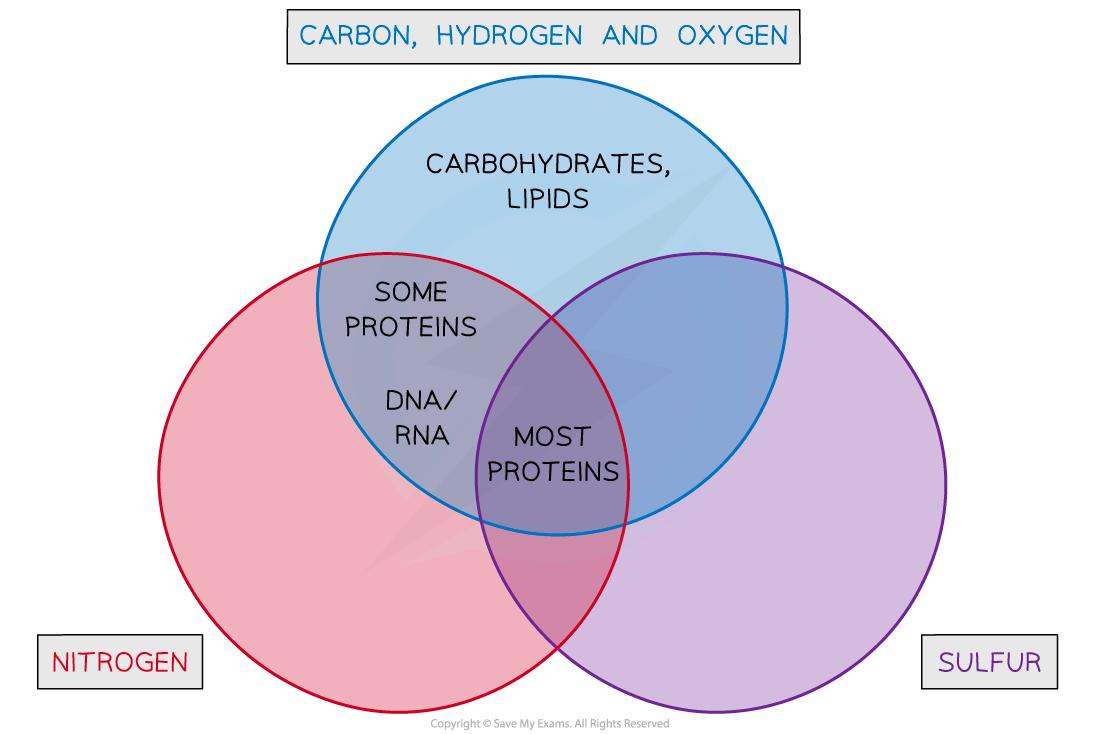
Using the Presence of Various Atoms to Identify Biochemicals
- Glycosidic bonds, ester bonds and peptide bonds all have a distinctive appearance in molecular drawings and will immediately identify carbohydrates, lipids and proteins respectively
Worked Example
Identify the Diagram (A, B, C or D) which Shows a Triglyceride Structure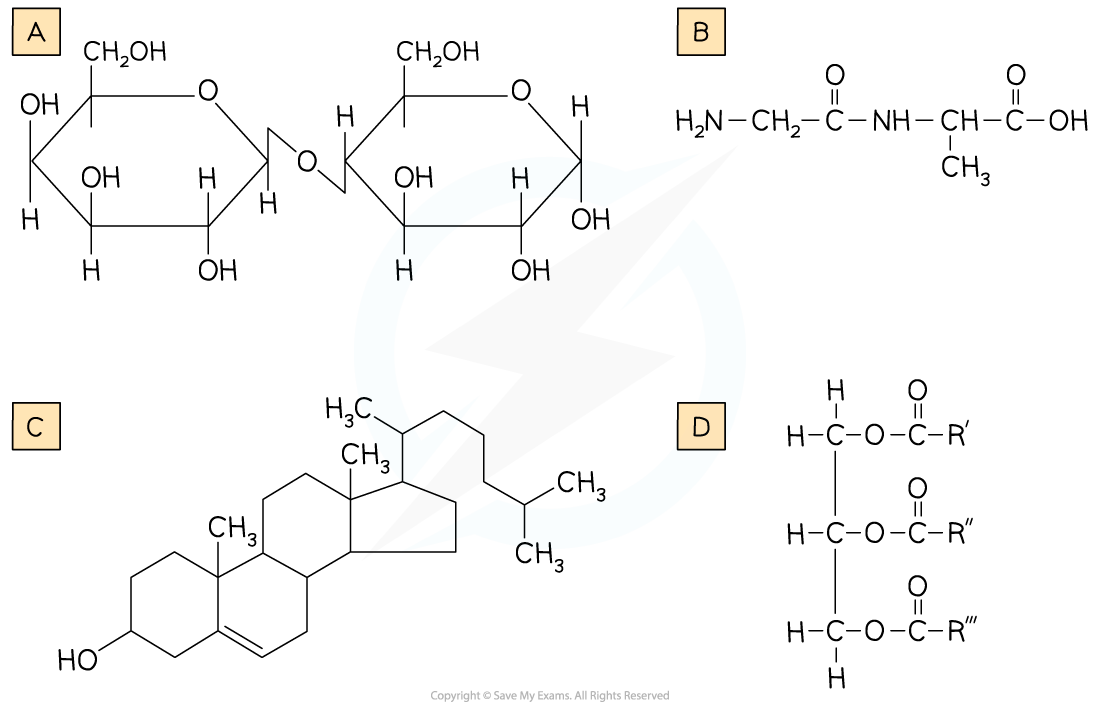
Step 1: Look at the elements present in all the diagrams
We're looking for a triglyceride, a type of lipid. Lipids contain C, H and O only. B also contains nitrogen (B is a dipeptide)
Eliminate Answer B
Step 2: Look for lipid structures
A contains ring structures, so is likely a carbohydrate (A is a disaccharide)
Eliminate Answer A
Step 3: Look for three hydrocarbon chains
D contains three hydrocarbon chains (attached by ester bonds to glycerol). C is cholesterol, which is a lipid, but not a triglyceride lipid.
Select Answer D
The ratio of hydrogen to oxygen
-
- The numbers of hydrogen and oxygen atoms in a molecule can help to identify it
- Carbohydrates contain hydrogen and oxygen in a 2:1 ratio
- Think of water, formula H2O, and where the '-hydrates' part of the word 'carbohydrates' comes from
- Lipids contain a much lower proportion of oxygen than carbohydrates eg. C18H34O2, where the hydrogen to oxygen ratio is 17:1
Molecular Diagrams: Peptide Bond Formation
- Having learned to draw the structure of a generalised amino acid, two or more of these can be joined together to show how peptide bonds form during protein synthesis
- Amino acid monomers link together via a condensation reaction
- This releases a molecule of water (H2O)
- One H atom (of the released water) comes from one amino acid's amine group
- The other H atom and an O atom come from the other amino acid's carboxylic acid group
- This knowledge can be useful when drawing how two amino acids condense to form a peptide bond
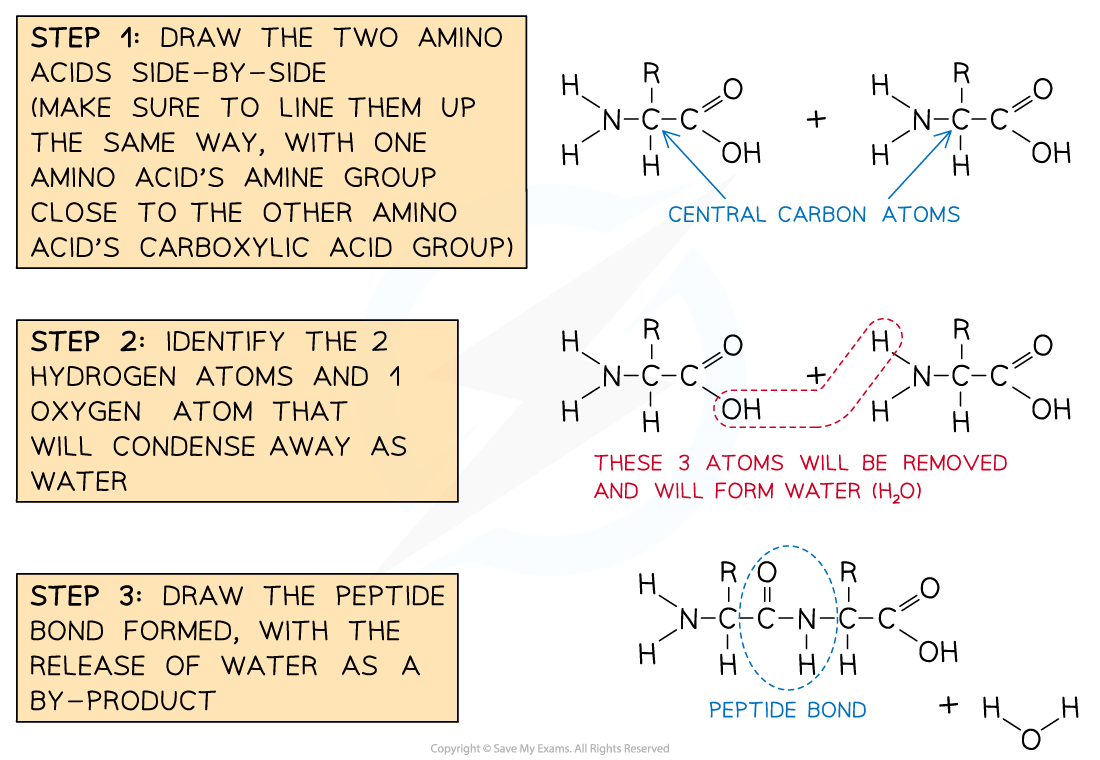
The recommended steps in drawing a peptide bond formation
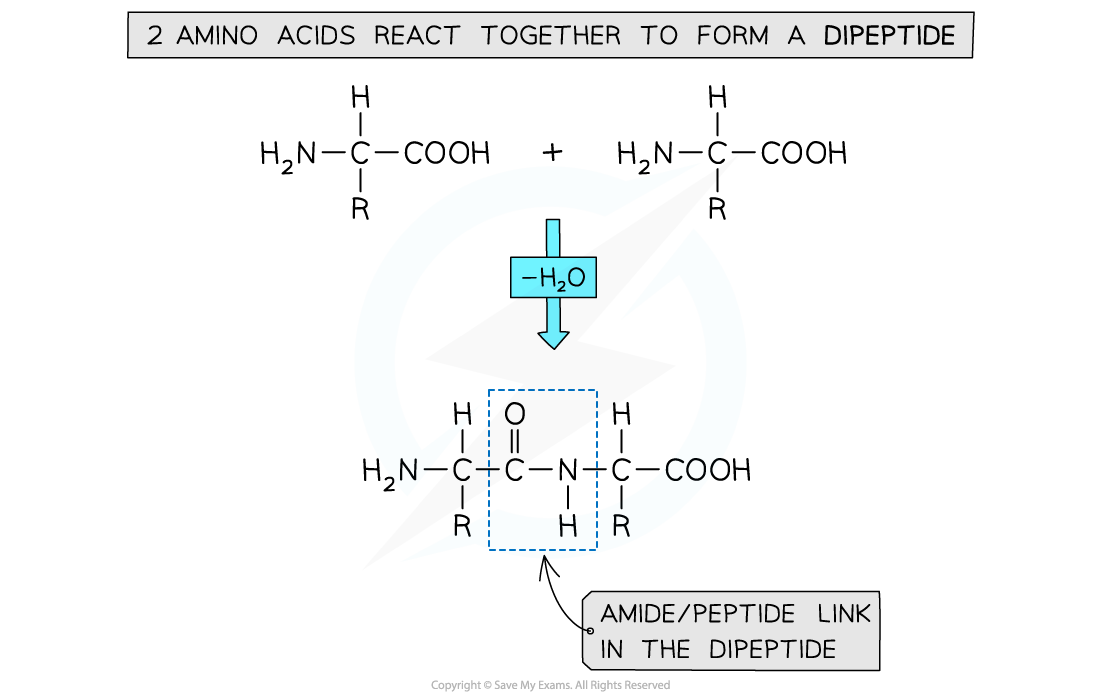
Formation of a dipeptide
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1