- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
IB DP Physics: HL复习笔记12.2.7 Measuring Half-Life
Measuring Half-Life
- Half-life is defined as:
The time taken for the initial number of nuclei to halve for a particular isotope
- This means when a time equal to the half-life has passed, the activity of the sample will also half
- This is because the activity is proportional to the number of undecayed nuclei, A ∝ N
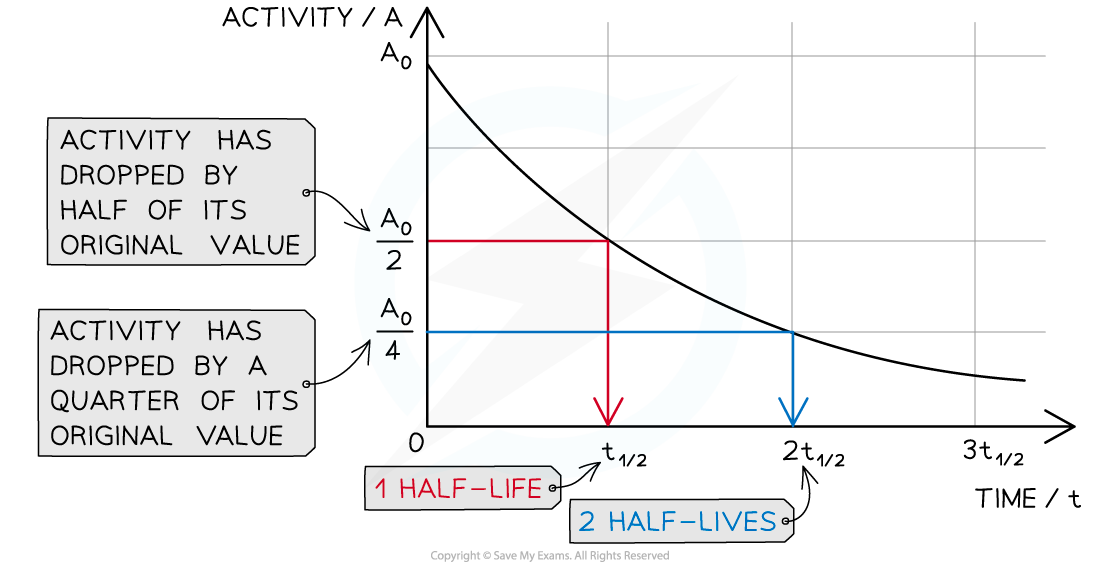
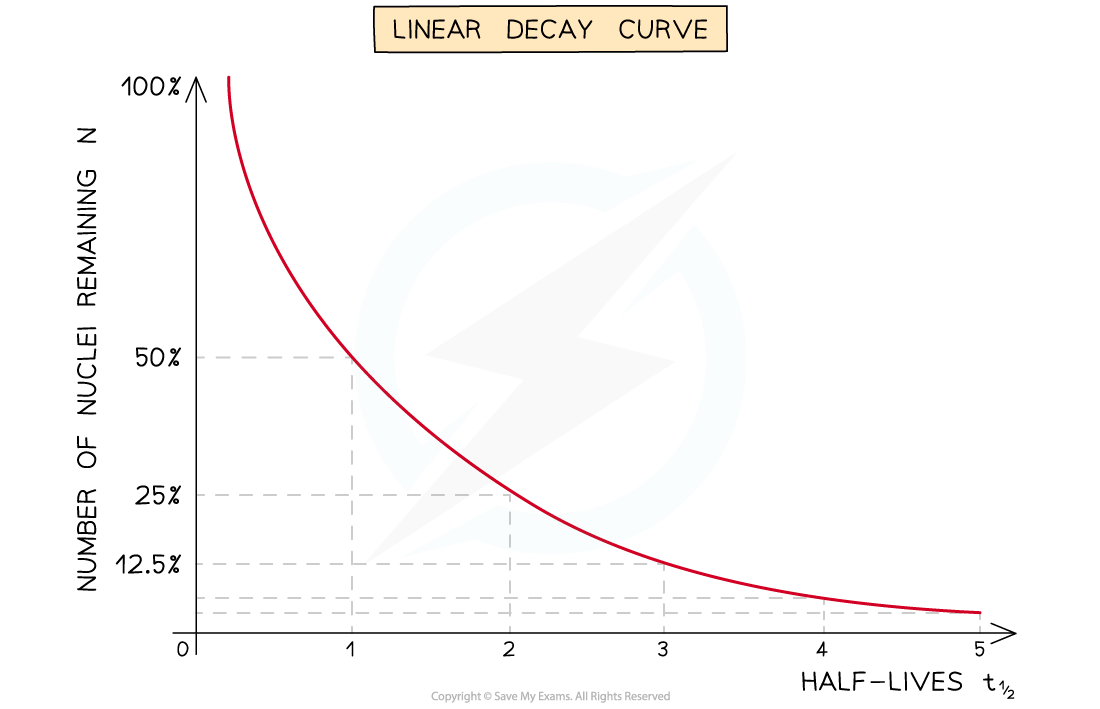
When a time equal to the half-life passes, the activity falls by half, when two half-lives pass, the activity falls by another half (which is a quarter of the initial value)
- To find an expression for half-life, start with the equation for exponential decay:
N = N0 e–λt
- Where:
- N = number of nuclei remaining in a sample
- N0 = the initial number of undecayed nuclei (when t = 0)
- λ = decay constant (s-1)
- t = time interval (s)
- When time t is equal to the half-life t½, the activity N of the sample will be half of its original value, so N = ½ N0
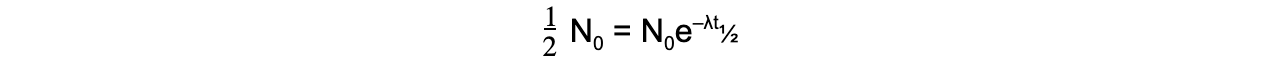
- The formula can then be derived as follows:
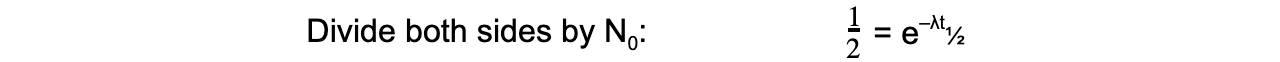


- Therefore, half-life t½ can be calculated using the equation:
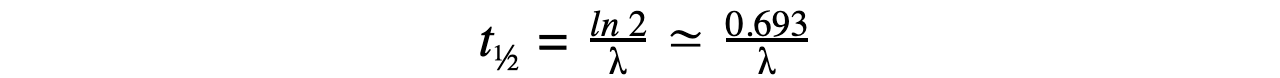
- This equation shows that half-life t½ and the radioactive decay rate constant λ are inversely proportional
- Therefore, the shorter the half-life, the larger the decay constant and the faster the decay
- The half-life of a radioactive substance can be determined from decay curves and log graphs
- Since half-life is the time taken for the initial number of nuclei, or activity, to reduce by half, it can be found by
- Drawing a line to the curve at the point where the activity has dropped to half of its original value
- Drawing a line from the curve to the time axis, this is the half-life
 Measuring Long Half-Lives
Measuring Long Half-Lives
- For nuclides with long half-lives, on the scale of years, this can be measured by:
- Measuring the mass of the nuclide in a pure sample
- Determining the number of atoms N in the sample using N = nNA
- Measuring the total activity A of the sample using the counts collected by a detector
 Note: The sample must be sufficiently large enough in order for a significant number of decays to occur per unit time so that an accurate measure of activity can be made
Note: The sample must be sufficiently large enough in order for a significant number of decays to occur per unit time so that an accurate measure of activity can be made
Measuring Short Half-Lives
- For nuclides with short half-lives, on the scale of seconds, hours or days, this can be measured by:
- Measuring the background count rate in the laboratory (to subtract from each reading)
- Taking readings of the count rate against time until the value equals that of the background count rate (i.e. until all of the sample has decayed)
- Plotting a graph of activity, A, against time, t (as corrected count rate ∝ activity, A)
- Making at least 3 estimates of half-life from the graph and taking a mean
OR
-
- Plotting a graph of ln N against time, t (as corrected count rate ∝ number of nuclei in the sample, N)
- Finding the gradient of this graph, which gives –λ
 Straight-line graphs tend to be more useful than curves for interpreting data
Straight-line graphs tend to be more useful than curves for interpreting data
-
- Due to the exponential nature of radioactive decay logarithms can be used to achieve a straight line graph
- Take the exponential decay equation for the number of nuclei
N = N0 e–λt
- Taking the natural logs of both sides
ln N = ln (N0) − λt
- In this form, this equation can be compared to the equation of a straight line
y = mx + c
- Where:
- ln (N) is plotted on the y-axis
- t is plotted on the x-axis
- gradient = −λ
- y-intercept = ln (N0)
- Half-lives can be found in a similar way to the decay curve but the intervals will be regular as shown below:
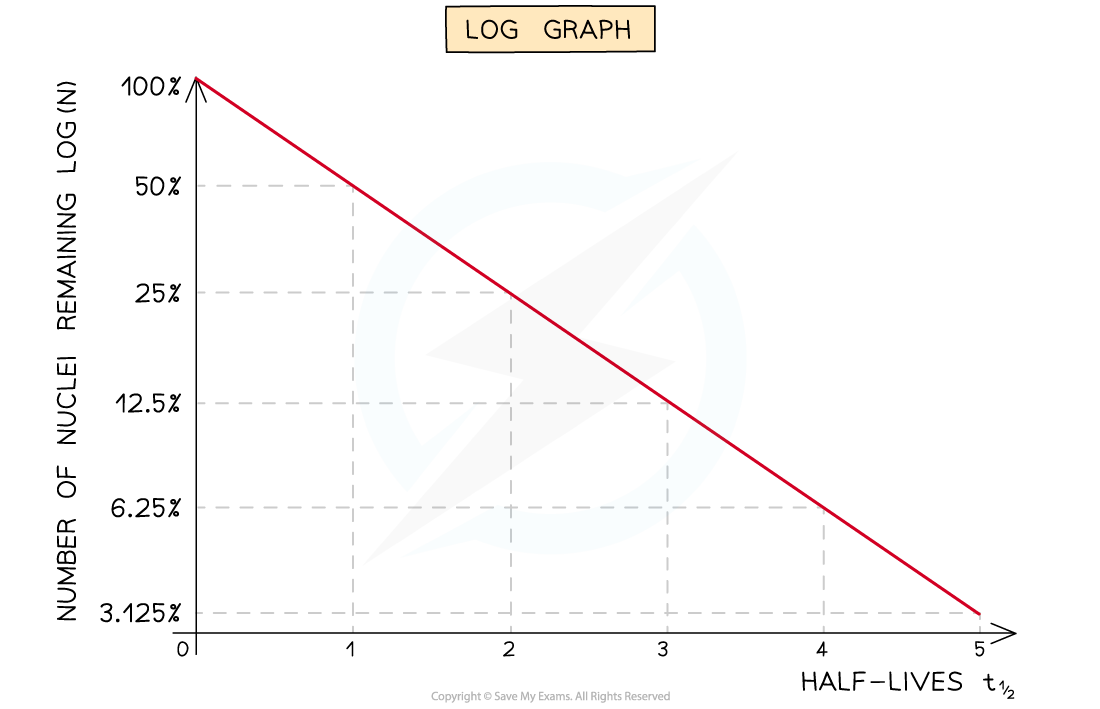
A logarithmic graph. This represents the relationship:
