- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
AQA A Level Chemistry复习笔记7.4.3 Friedel–Crafts Acylation
Friedel–Crafts Acylation
- Arenes are very stable compounds due to the delocalisation of π electrons in the ring
- This is because the negative charge is spread out over the molecule instead of being confined to a small area
- During chemical reactions such as substitution reactions, this delocalised ring is maintained
- Addition reactions however, disrupt the aromatic stabilisation
- Arenes undergo a series of reactions including:
- Substitution
- Nitration
- Friedel-Crafts alkylation
- Friedel-Crafts acylation
- Hydrogenation
- Alkylarenes such as methylbenzene undergo halogenation on the 2 or 4 positions
- This is due to the electron-donating alkyl groups which activate these positions
- Phenol (C6H5OH) and phenylamine (C6H5NH2) are also activated in the 2 and 4 positions
- The halogenation of alkylarenes therefore result in the formation of two products
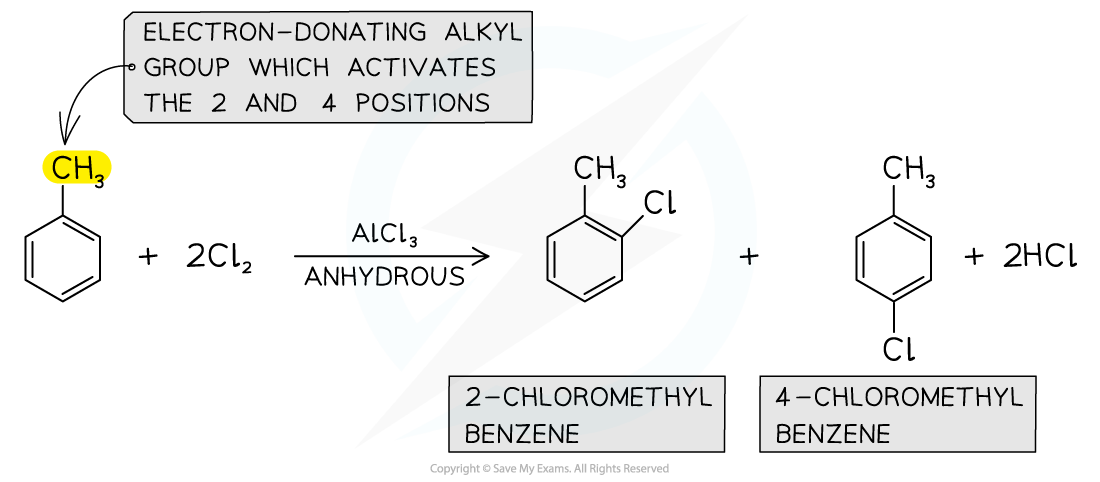
Alkylarenes are substituted on the 2 or 4 position
- Multiple substitutions occur when excess halogen is used
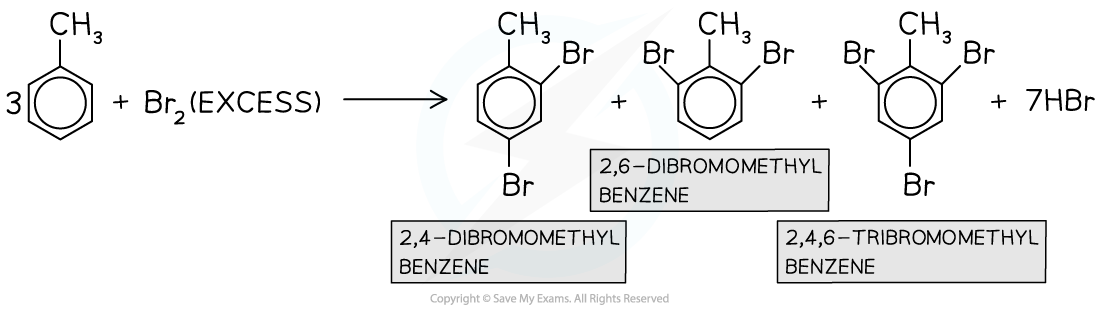
In the presence of excess halogen, multiple substitutions occur
- Again, due to the electron-donating alkyl groups in alkylarenes, nitration of methylbenzene will occur on the 2 and 4 position
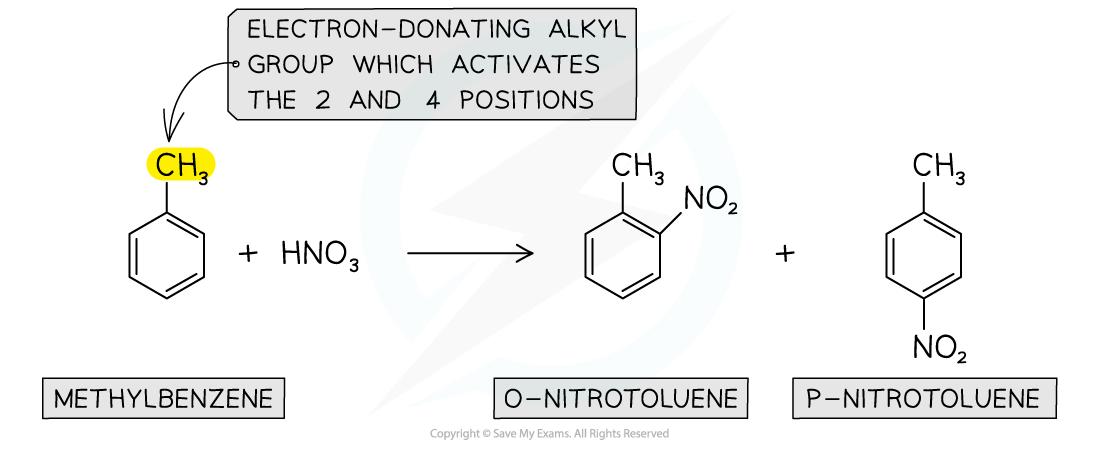
Nitration of alkylarenes
Friedel-Crafts Reactions
- Friedel-Crafts reactions are also electrophilic substitution reactions
- Due to the aromatic stabilisation in arenes, they are often unreactive
- To use arenes as starting materials for the synthesis of other organic compounds, their structure, therefore, needs to be changed to turn them into more reactive compounds
- Friedel-Crafts reactions can be used to substitute a hydrogen atom in the benzene ring for an alkyl group (Friedel-Crafts alkylation) or an acyl group (Friedel-Crafts acylation)
- Like any other electrophilic substitution reaction, the Friedel-Crafts reactions consist of three steps:
- Generating the electrophile
- Electrophilic attack on the benzene ring
- Regenerating aromaticity of the benzene ring
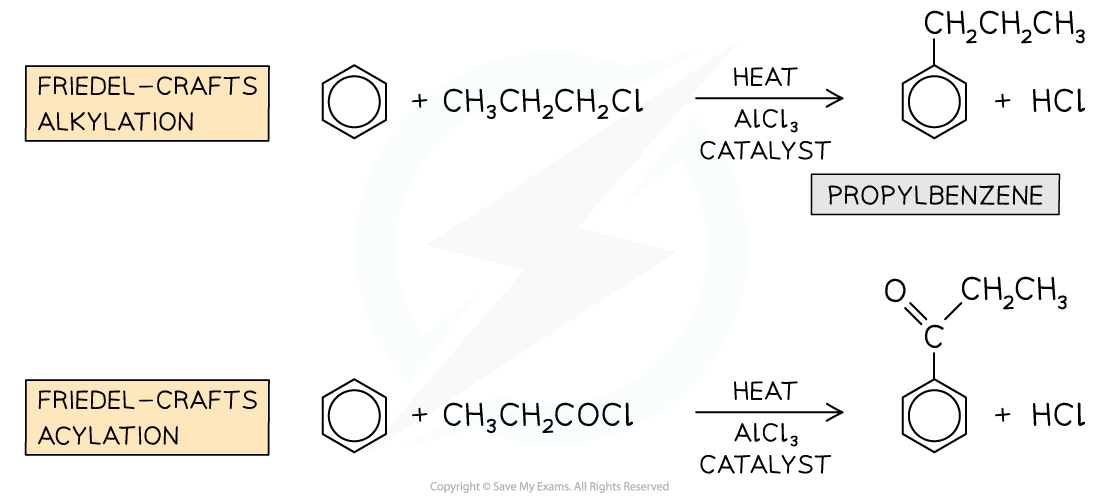
Examples of Friedel-Crafts alkylation and acylation reactions
Friedel-Crafts Acylation
- In the Friedel-Crafts acylation reaction, an acyl group is substituted into the benzene ring
- An acyl group is an alkyl group containing a carbonyl, C=O group
- The benzene ring is reacted with an acyl chloride in the presence of an AlCl3 catalyst
- An example of an acylation reaction is the reaction of methylbenzene with propanoyl chloride to form an acyl benzene
- Note that the acyl group is on the 4 position due to the -CH3 group on the benzene
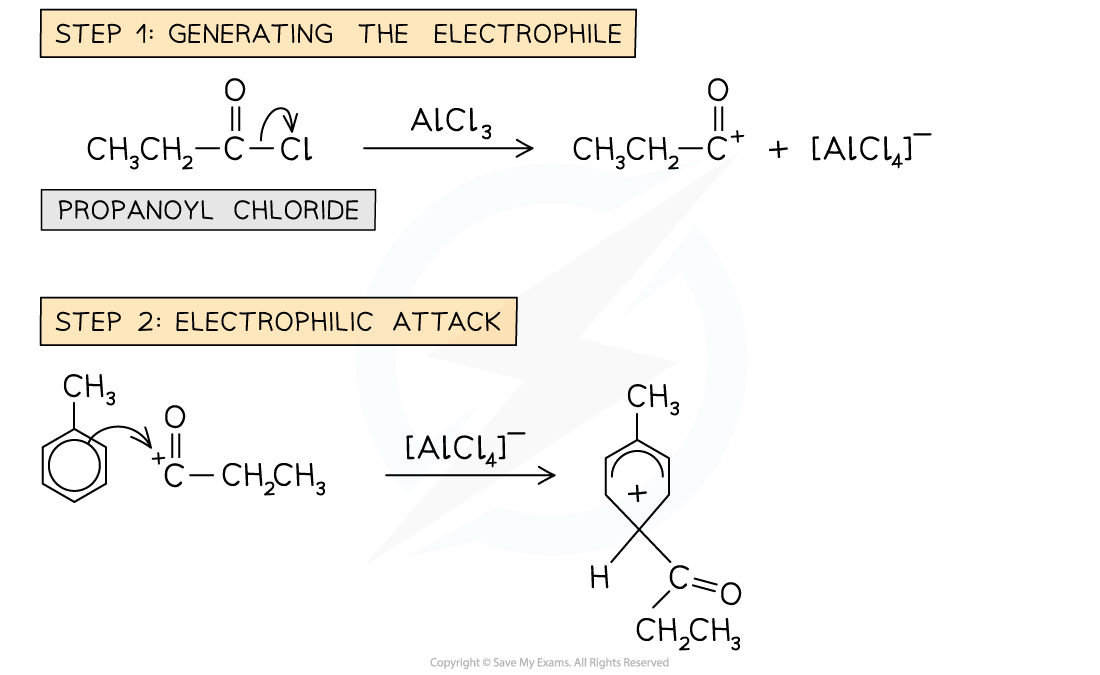
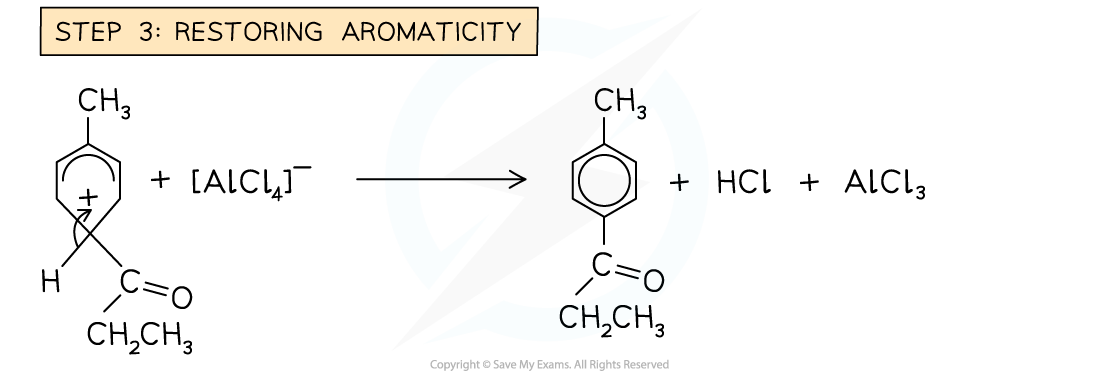
Example of a Friedel-Crafts acylation reaction
Hydrogenation
- The hydrogenation of benzene is an addition reaction
- Benzene is heated with hydrogen gas and a nickel or platinum catalyst to form cyclohexane

Hydrogenation of benzene
- The same reaction occurs when ethylbenzene undergoes hydrogenation to form cycloethylbenzene

Hydrogenation of ethylbenzene
Summary of Reactions of Arenes Table
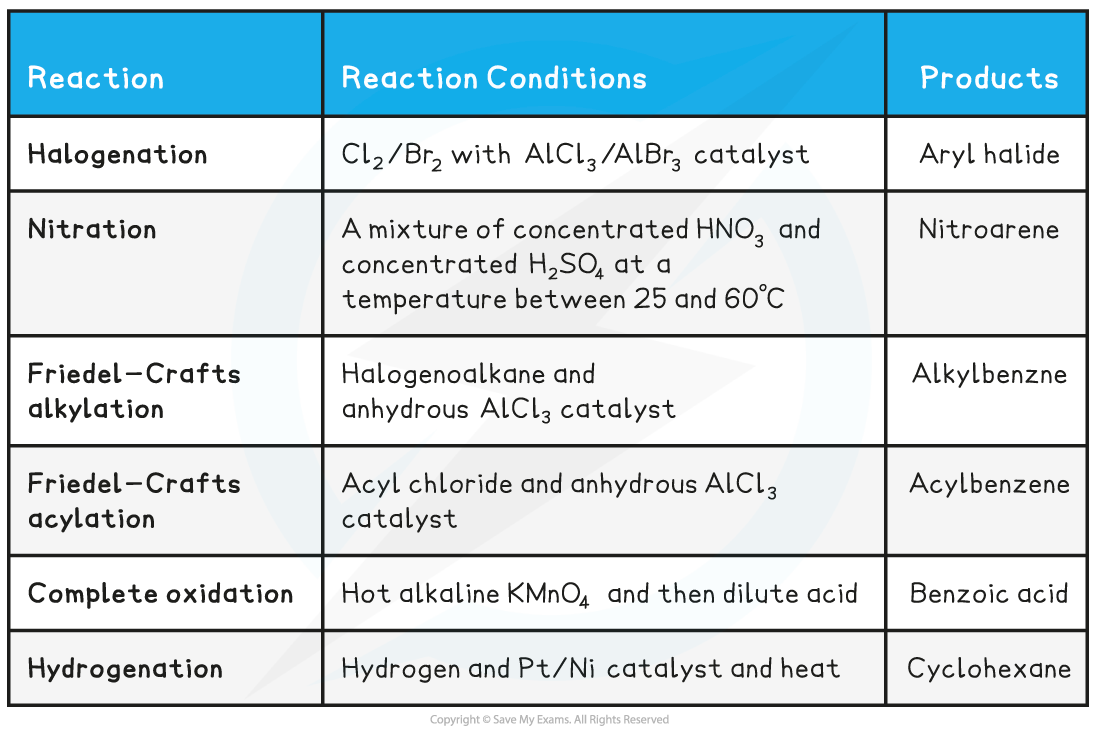
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





