- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
AQA A Level Chemistry复习笔记1.2.2 Empirical & Molecular Formula
Empirical & Molecular Formulae
- The molecular formula is the formula that shows the number and type of each atom in a molecule
- E.g. the molecular formula of ethanoic acid is C2H4O2
- The empirical formula is the simplest whole number ratio of atoms of each element present in one molecule or formula unit of a compound
- E.g. the empirical formula of ethanoic acid is CH2O
Empirical & Molecular Formulae Calculations
Empirical formula
- Empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound
- It is calculated from knowledge of the ratio of masses of each element in the compound
- The empirical formula can be found by determining the mass of each element present in a sample of the compound
- It can also be deduced from data that gives the percentage compositions by mass of the elements in a compound
Worked Example
Empirical formula from mass
Determine the empirical formula of a compound that contains 10 g of hydrogen and 80 g of oxygen.
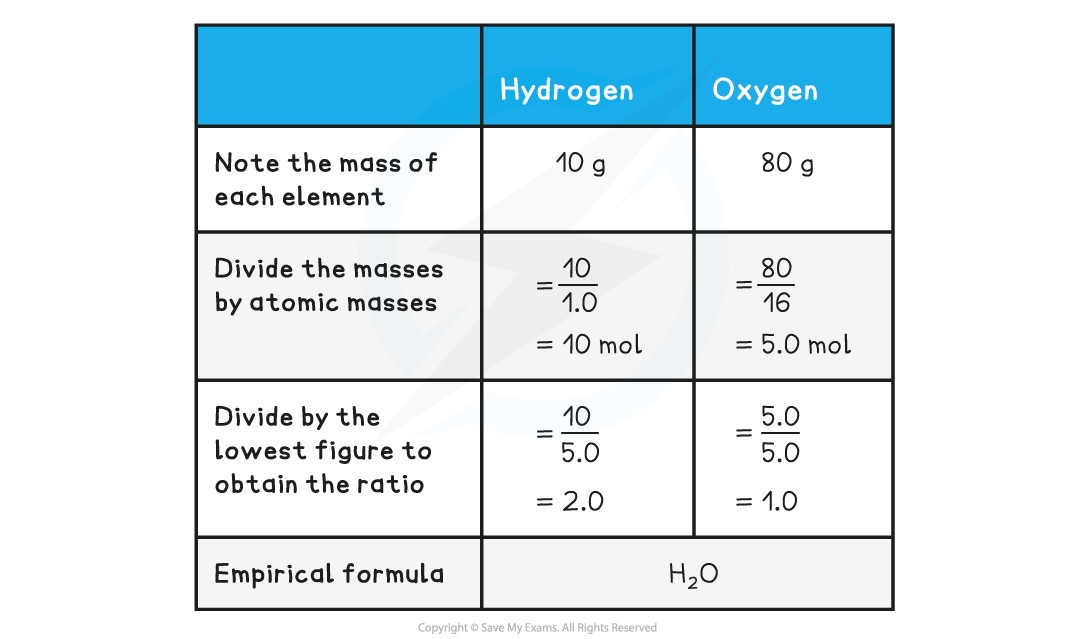
- The above example shows how to calculate empirical formula from the mass of each element present in the compound
- The example below shows how to calculate the empirical formula from percentage composition
Worked Example
Empirical formula from %
Determine the empirical formula of a compound that contains 85.7% carbon and 14.3% hydrogen.

Molecular formula
- The molecular formula gives the exact numbers of atoms of each element present in the formula of the compound
- The molecular formula can be found by dividing the relative formula mass of the molecular formula by the relative formula mass of the empirical formula
- Multiply the number of each element present in the empirical formula by this number to find the molecular formula
Worked Example
Calculating molecular formula
The empirical formula of X is C4H10S and the relative molecular mass of X is 180
What is the molecular formula of X?
(Ar data: C = 12, H = 1, S = 32)
Answer
Step 1: Calculate relative mass of the empirical formula
-
- Relative empirical mass = (C x 4) + (H x 10) + (S x 1)
- Relative empirical mass = (12 x 4) + (1 x 10) + (32 x 1)
- Relative empirical mass = 90
Step 2: Divide relative formula mass of X by relative empirical mass
-
- Ratio between Mr of X and the Mr of the empirical formula = 180/90
- Ratio between Mr of X and the Mr of the empirical formula = 2
Step 3: Multiply each number of elements by 2
-
- (C4 x 2) + (H10 x 2) + (S x 2) = (C8) + (H20) + (S2)
- Molecular Formula of X is C8H20S2
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





