- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel IGCSE Biology 复习笔记 2.5.1 Photosynthesis
Edexcel IGCSE Biology 复习笔记 2.5.1 Photosynthesis
Photosynthesis Theory
- Photosynthesis is an endothermic reaction in which energy from sunlight is transferred to the chloroplasts in green plants
- Green plants use this energy to make the carbohydrate glucose from the raw materials carbon dioxide and water
- At the same time, oxygen is made and released as a waste product
- Photosynthesis can be defined as the process by which plants manufacture carbohydrates from raw materials using energy from light
- Plants are
- Autotrophs – they can make complex molecules (glucose) from simple molecules (carbon dioxide and water)
- Producers - they can make their own food and so are the first organism at the start of all food chains
The products of photosynthesis
- Plants use the glucose they make as a source of energy in respiration
- They can also use it to
- Produce starch for storage
- Synthesise lipids for an energy source in seeds
- To form cellulose to make cell walls
- Produce amino acids (used to make proteins) when combined with nitrogen and other mineral ions absorbed by roots
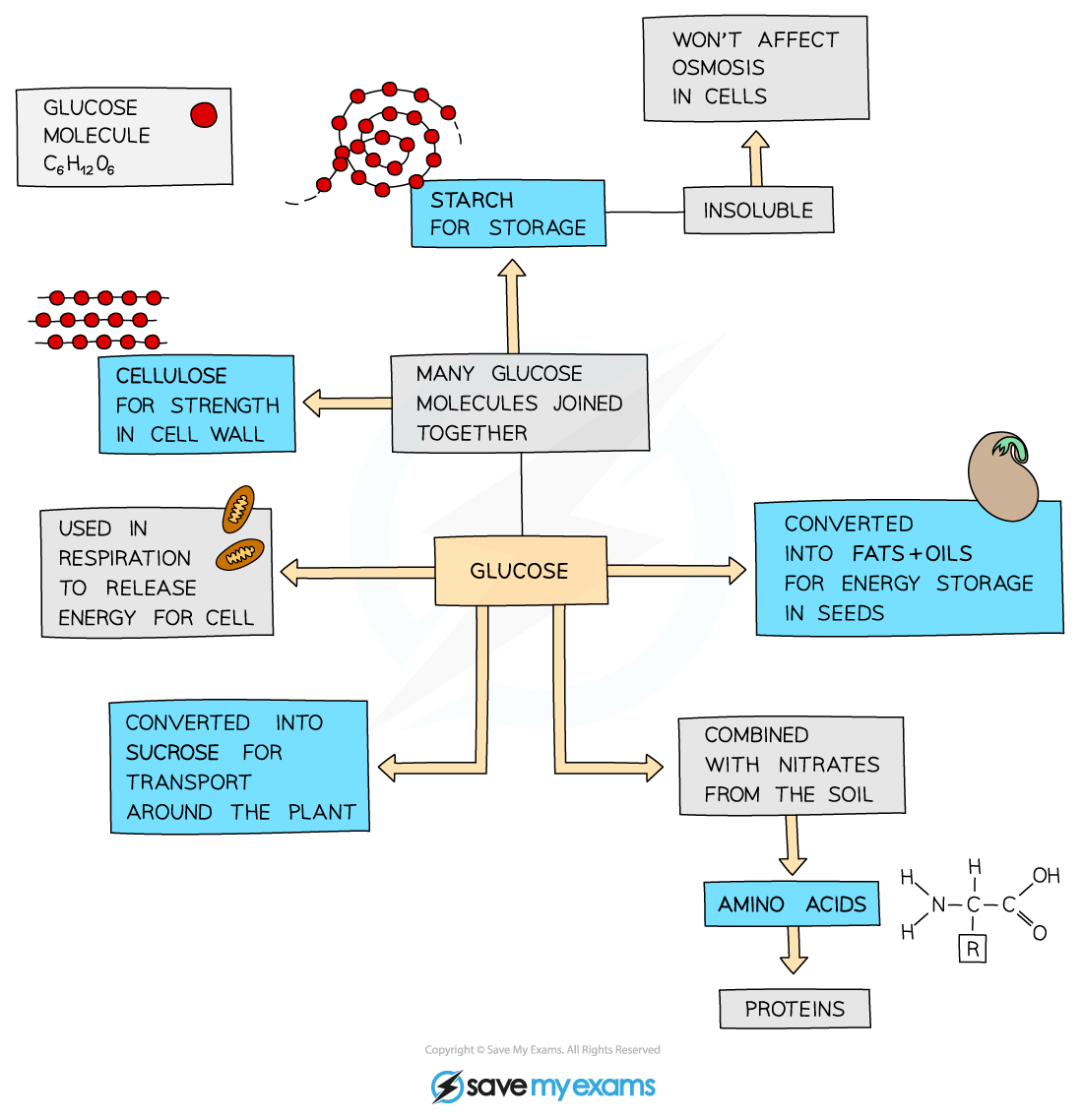
The fate of glucose produced in photosynthesis
Exam Tip
If asked for the raw materials required for photosynthesis, the answer is carbon dioxide and water.Although required for the reaction to take place, light energy is not a substance and therefore cannot be a raw material.
Photosynthesis Equation
- Photosynthesis can be summarised in a word equation as shown below:
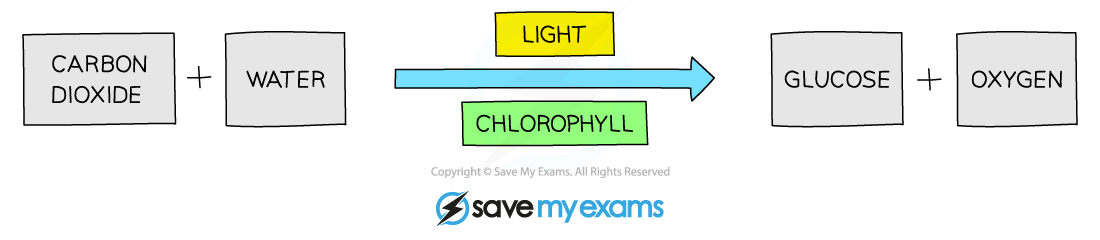
Word equation for photosynthesis
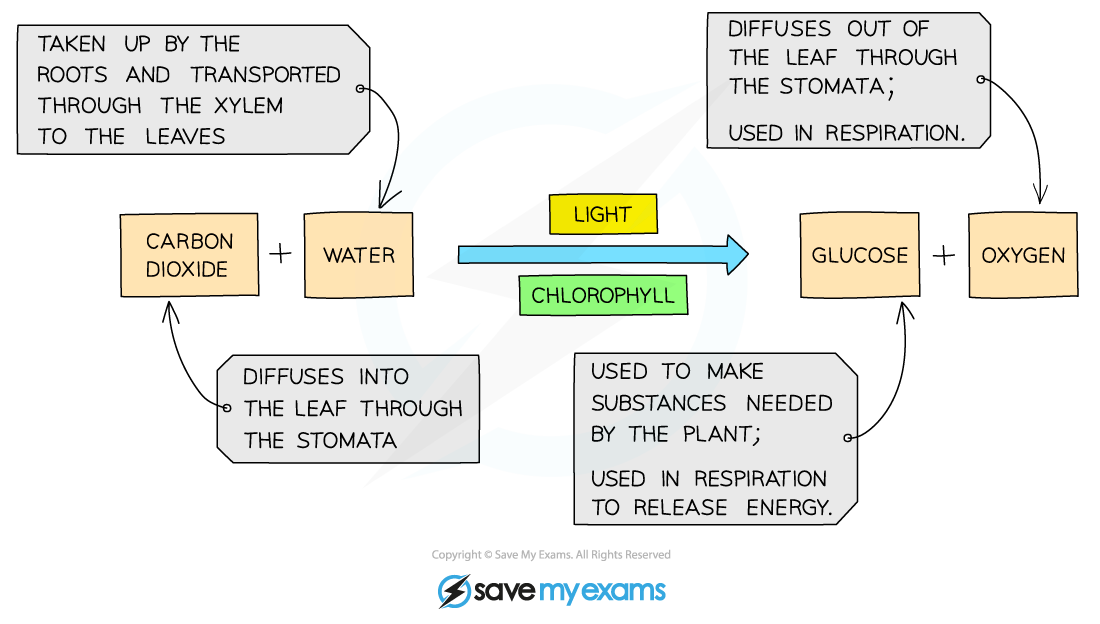
Where do the reactants come from and where do the products go?
- This equation can also be shown as a balanced chemical equation
- Six carbon dioxide molecules combine with six water molecules to make one glucose molecule and six oxygen molecules

The balanced chemical equation for photosynthesis
Exam Tip
The photosynthesis equation is the exact reverse of the aerobic respiration equation so if you have learned one you also know the other one! You will usually get more marks for providing the balanced chemical equation than the word equation.
转载自savemyexam
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





