- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
AQA A Level Biology复习笔记2.5.8 Antibodies
Antibodies: Structure
Structure
- Antibodies are globular glycoproteins called immunoglobulins
- Antibodies have a quaternary structure (which is represented as Y-shaped), with two ‘heavy’ (long) polypeptide chains bonded by disulfide bonds to two ‘light’ (short) polypeptide chains
- Each polypeptide chain has a constant region and variable region
- The constant regions do not vary within a class (isotype) of antibodies but do vary between the classes. The constant region determines the mechanism used to destroy the antigens
- There are 5 classes of mammalian antibodies each with different roles
- The amino acid sequence in the variable regions of the antibodies (the tips of the "Y") are different for each antibody. The variable region is where the antibody attaches to the antigen to form an antigen-antibody complex
- At the end of the variable region is a site called the antigen-binding site. Each antigen-binding site is generally composed of 110 to 130 amino acids and includes both the ends of the light and heavy chains
- The antigen-binding sites vary greatly giving the antibody its specificity for binding to antigens. The sites are specific to the epitope (the part of the antigen that binds to the antibody)
- A pathogen or virus may therefore present multiple antigens different antibodies need to be produced
- The ‘hinge’ region (where the disulfide bonds join the heavy chains) gives flexibility to the antibody molecule which allows the antigen-binding site to be placed at different angles when binding to antigens
- This region is not present in all classes of antibodies
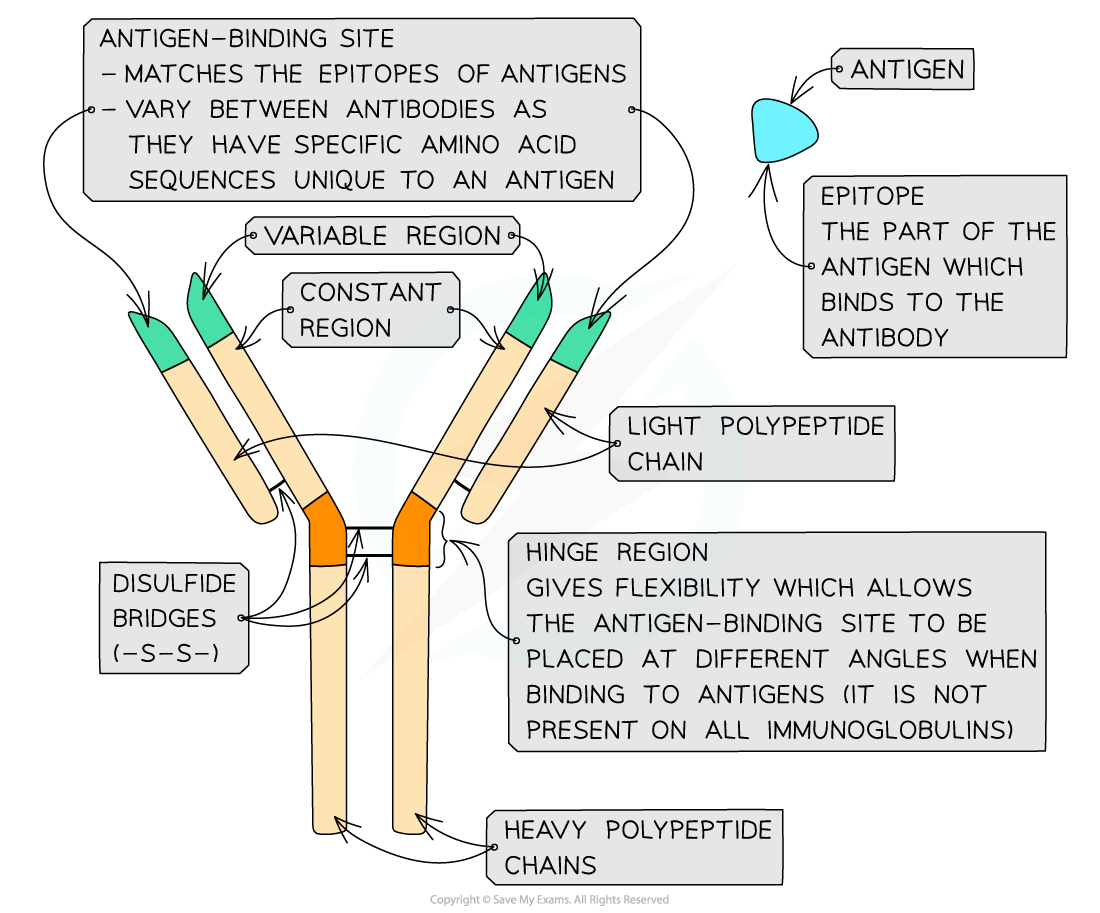
A model of the generalised structure of an antibody molecule
Exam Tip
You must know that each antibody will have a different variable region with an antigen-binding site that matches one antigen or toxin produced by a pathogen. The antigen-binding site (and therefore the antibody) is specific to one antigen.
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





