- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
AQA A Level Biology复习笔记1.2.5 Lipid Diagrams & Properties
Lipid Diagrams & Properties
- Triglycerides are a form of lipid
- They are made up of one molecule of glycerol with three fatty acids attached to it
- These fatty acids have long hydrocarbon ‘tails’
- Fatty acids occur in two forms:
- saturated fatty acids
- unsaturated fatty acids
- The difference between these fatty acid types is found in their hydrocarbon tails
Saturated fatty acids
- In saturated fatty acids, the bonds between the carbon atoms in the hydrocarbon tail are all single bonds
- The fatty acid is said to be ‘saturated’ with hydrogen
- This means that each carbon atom in the hydrocarbon tail (except for the final carbon atom) is bonded to two hydrogen atoms
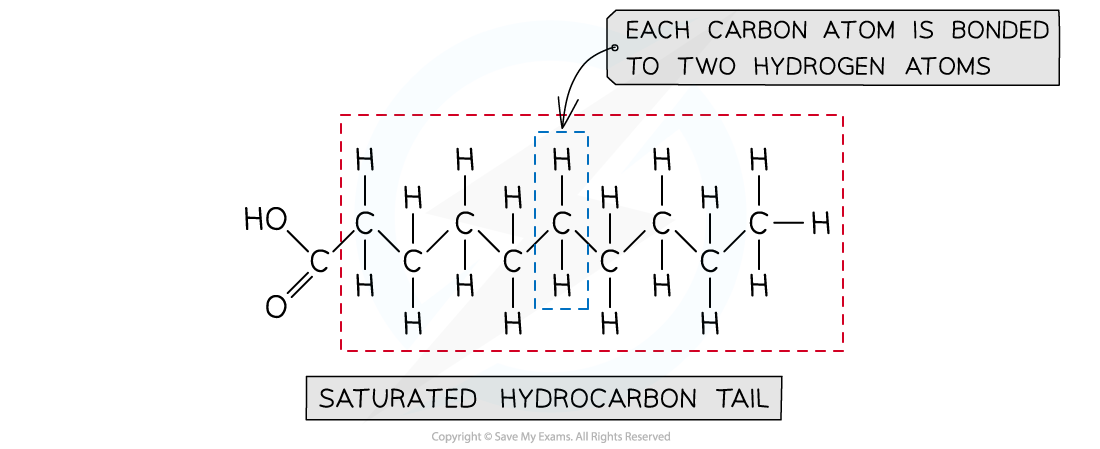
An example of a saturated fatty acid
Unsaturated fatty acids
- In unsaturated fatty acids, the bonds between the carbon atoms in the hydrocarbon tail are not all single bonds
- There is at least one carbon-carbon double bond (in some unsaturated fatty acids there are many carbon-carbon double bonds)
- These double bonds cause the hydrocarbon tail of unsaturated fatty acids to kink (bend slightly), meaning they are not as straight as saturated fatty acids
- The fatty acid is said to be ‘unsaturated’ because the hydrocarbon tail does not contain the maximum number of hydrogen atoms possible
- This is because each carbon atom in a carbon-carbon double bond can only bond to one hydrogen atom (instead of two)
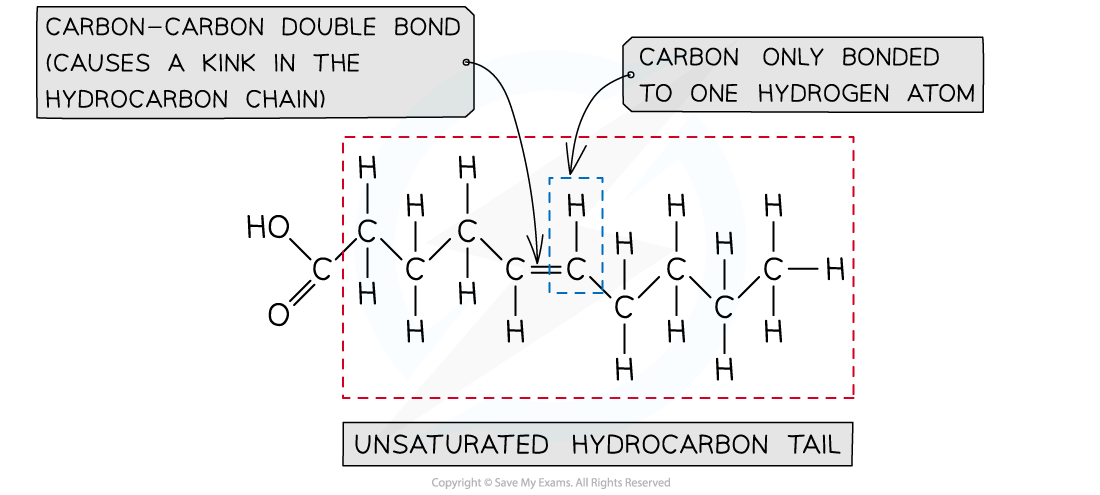
An example of an unsaturated fatty acid
Properties of triglycerides
- Triglycerides are mainly used as energy storage molecules
- This is because the long hydrocarbon tails of the fatty acids in triglycerides contain large amounts of chemical energy, which can be released when the fatty acids are broken down
- Triglycerides are also suitable as energy storage molecules because they are insoluble, meaning that they don’t affect the water potential inside the cell
- Inside cells, triglycerides form insoluble droplets, with the hydrophobic (water-repelling) fatty acids on the inside and the glycerol molecules on the outside
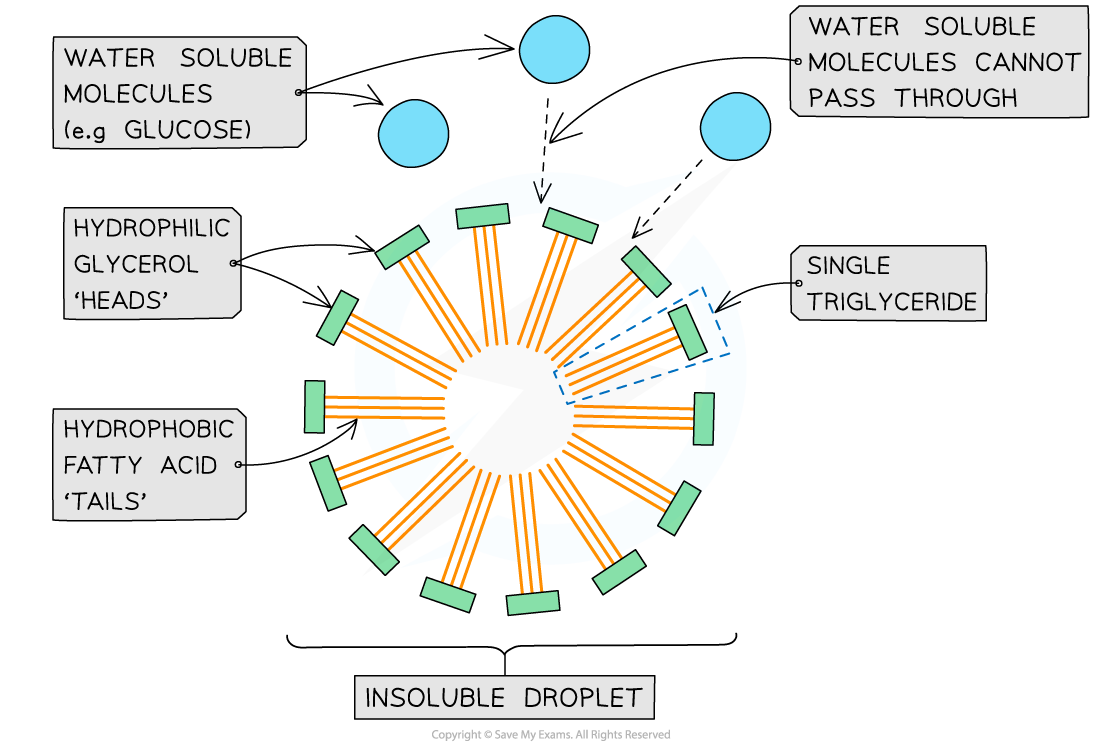
Triglycerides are suitable as energy storage molecules as they form insoluble droplets inside cells
Properties of phospholipids
- Phospholipids are another kind of lipid
- Phospholipids are similar in structure to triglycerides
- In phospholipids, one of the three fatty acid molecules attached to glycerol is replaced by a phosphate group
- This phosphate group is hydrophilic (water-loving), whereas the two fatty acids are hydrophobic (like in triglycerides)
- This makes phospholipids suitable for making up the bilayer of cell membranes, with the fatty acids facing inwards and the phosphate groups facing outwards
- This is also useful as it means the centre of the phospholipid bilayer is hydrophobic, meaning water-soluble substances cannot easily pass through
- This allows the cell membrane to act as a barrier, controlling what substances enter and leave the cell
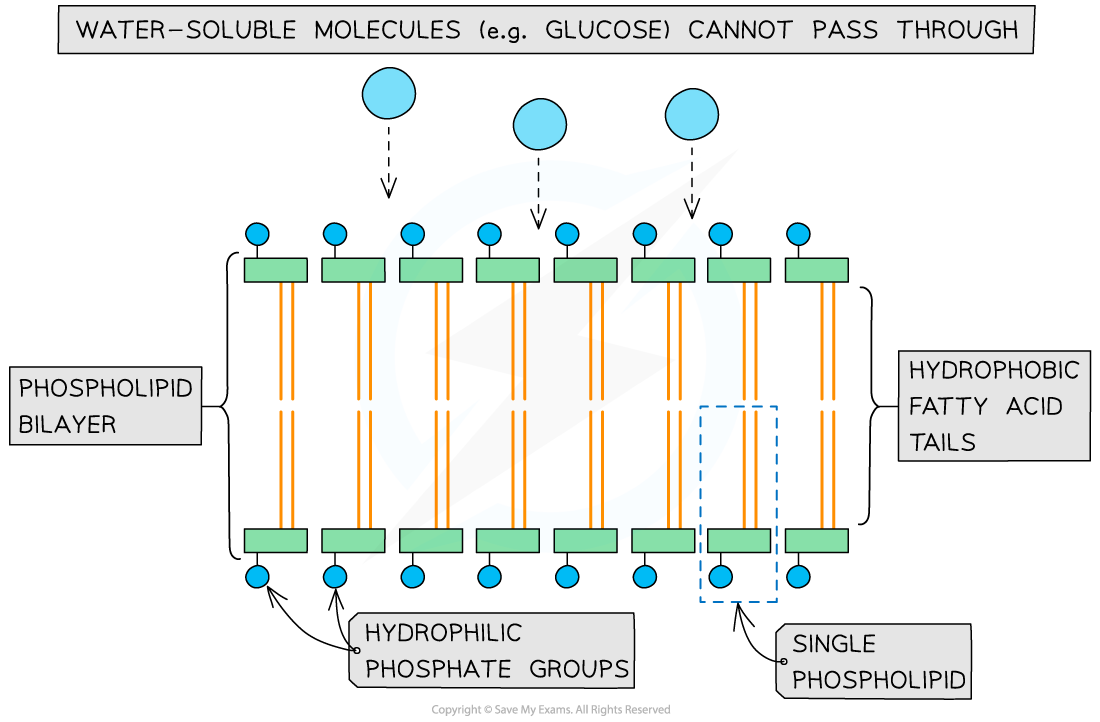
Phospholipids are suitable for making up cell membranes as they form a bilayer
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





