- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
AQA A Level Chemistry复习笔记3.4.1 Structure, Bonding & Reactivity
Shapes & Bond Angles in Organic Molecules
- Each carbon atom has four electrons in its outer shell (electronic configuration: 1s22s22p2)
- Carbon atoms share these four electrons in four covalent bonds with other atoms to achieve a full outer shell configuration
- These electrons are found in orbitals within the respective atoms
- When forming a covalent bond, the orbitals overlap in such a way to form two types of bonds
- Sigma bonds (σ)
- Pi bonds (π)
- When carbon atoms use only three of their electron pairs to form a σ bond, each carbon atom will have a p orbital which contains one spare electron
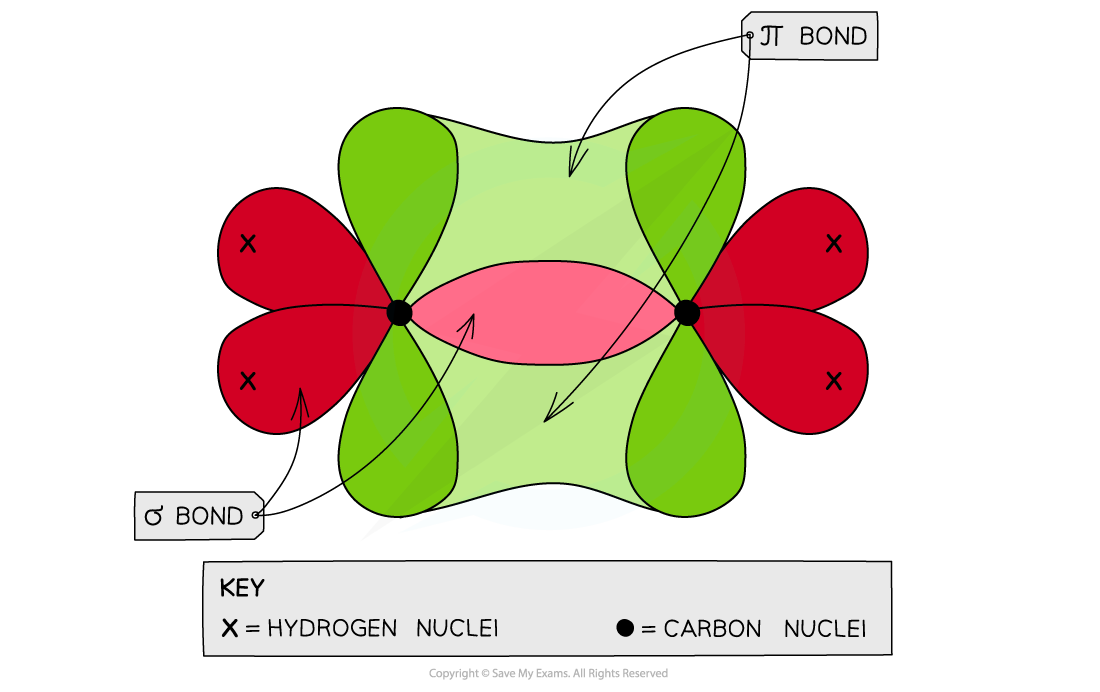
- When the p orbitals of two carbon atoms overlap with each other, a π bond is formed (the π bond contains two electrons)
- The two orbitals that form the π bond lie above and below the plane of the two carbon atoms to maximise bond overlap
- The three bonding pair of electrons are in the plane of the molecule and repel each other
- The molecule adopts a planar arrangement with bond angles of 120o
Ethene
-
- Each carbon atom uses three of its four electrons to form σ bonds
- Two σ bonds are formed with the hydrogen atoms
- One σ bond is formed with the other carbon atom
- The fourth electron from each carbon atom occupies a p orbital which overlaps sideways with another p orbital on the other carbon atom to form a π bond
- This means that the C-C is a double bond: one σ and one π bond
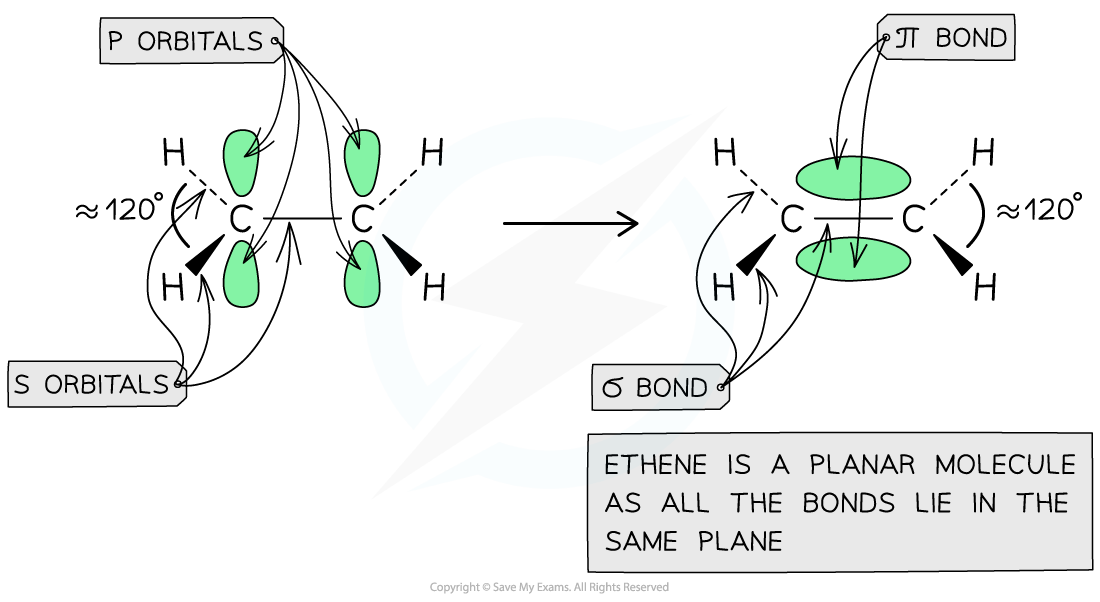
The overlap of the two p orbitals results in the formation of a π bond in ethene in which the bonding pair of electrons repel each other to force the molecule into a planar configuration with bond angles of 120 o
Each carbon atom in ethene forms two sigma bonds with hydrogen atoms and one σ bond with another carbon atom. The fourth electron is used to form a π bond between the two carbon atoms
Exam Tip
A double bond is a combination of a σ and π bond and a triple bond is a combination of one σ and two π bonds. The strength of the bonds increases as follows: single < double < triple bond This is due to the increased electron density around the carbon-carbon bond, making the bond stronger and more difficult to break. You must be able to draw and explain these diagrams for the exam.
Molecular Orbitals
Bond overlap in covalent bonds
- A single covalent bond is formed when two nonmetals combine
- Each atom that combines has an atomic orbital containing a single unpaired electron
- When a covalent bond is formed, the atomic orbitals overlap to form a combined orbital containing two electrons
- This new orbital is called the molecular orbital
- The greater the atomic orbital overlap, the stronger the bond
- Sigma (σ) bonds are formed by end-to-end overlap of orbitals between the bonding atoms
- Pi (π) bonds are formed by the sideways overlap of adjacent above and below the σ bond
σ bonds
- Sigma (σ) bonds are formed from the end to end overlap of atomic orbitals
- s orbitals overlap this way as well as p orbitals

Sigma orbitals can be formed from the end to end overlap of s orbitals
- The electron density in a σ bond is symmetrical about a line joining the nuclei of the atoms forming the bond
- The pair of electrons is found between the nuclei of the two atoms
- The electrostatic attraction between the electrons and nuclei bonds the atoms to each other
Hydrogen
- The hydrogen atom has only one s orbital
- The s orbitals of the two hydrogen atoms will overlap to form a σ bond

π bonds
- Pi (π) bonds are formed from the sideways overlap of adjacent p orbitals
- The two lobes that make up the π bond lie above and below the plane of the σ bond
- This maximises overlap of the p orbitals
- A single π bond is drawn as two electron clouds, one arising from each lobe of the p orbitals
- The two clouds of electrons in a π bond represent one bond containing two electrons
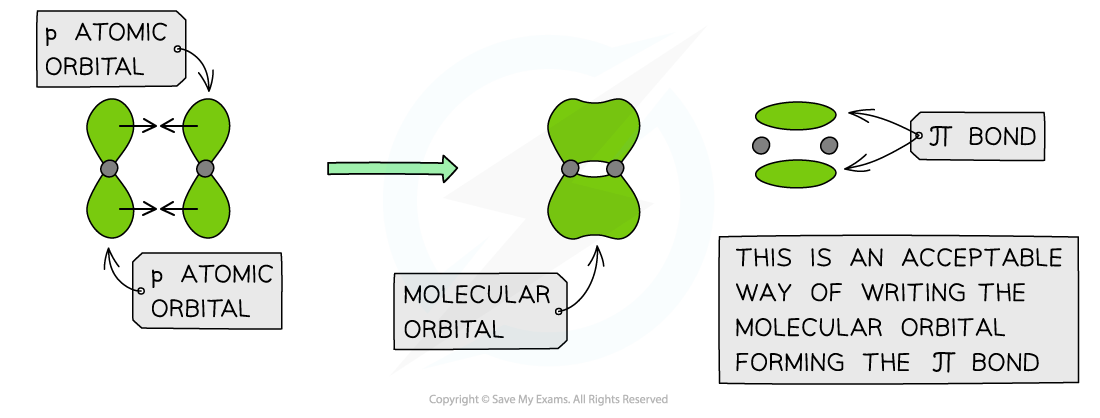
π orbitals can be formed from the sideways overlap of p orbitals
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





