- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
AQA A Level Chemistry复习笔记3.1.1 Functional Groups
Organic Definitions & Terminology
- Organic chemistry is the chemistry of carbon compounds
- Carbon forms a vast number of compounds because it can form strong covalent bonds with itself
- This enables it to form long chains of carbon atoms, and hence an almost infinite variety of carbon compounds are known

An almost infinite variety of chains, branches and rings is possible
Homologous series
- A homologous series is a group of organic compounds that have the same functional group, but each successive member differs by CH2
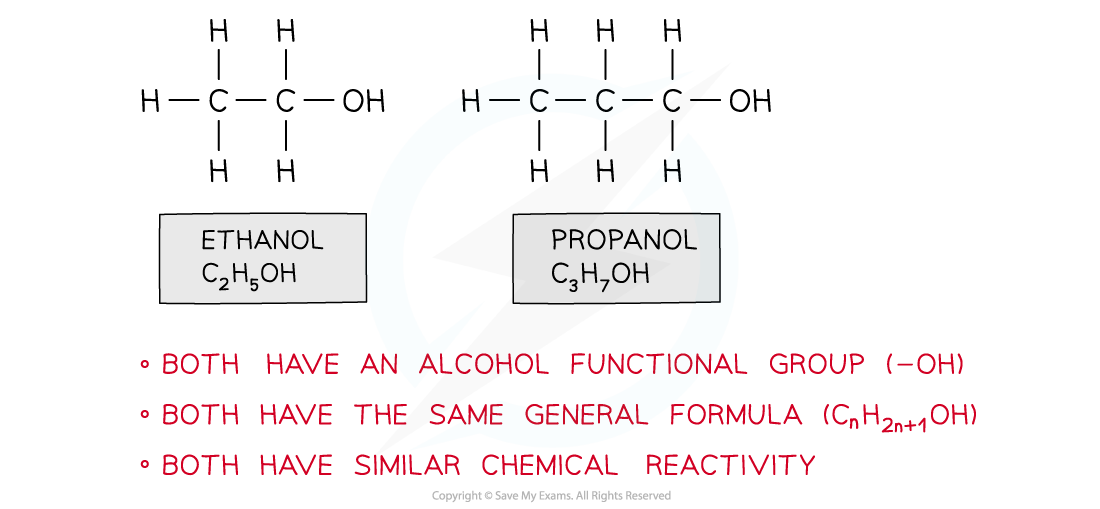
Ethanol and propanol belong to the same homologous series
Homologous Series of Alkanes
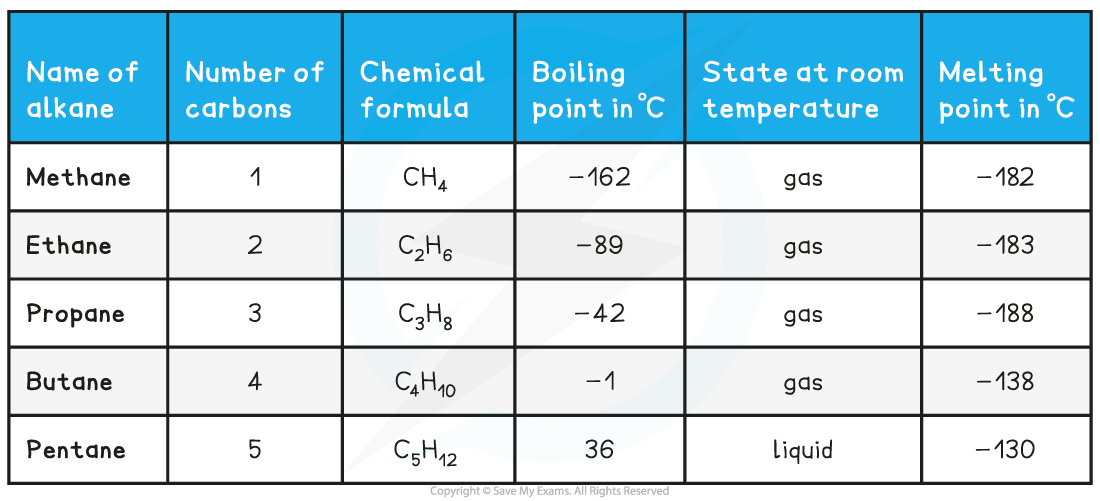
- Things we can say about a homologous series:
- Each member has the same functional group
- Each member has the same general formula
- Each member has similar chemical properties
- Each successive member differs by -CH2 –
- Members have gradually changing physical properties, for example, boiling point, melting point and density
- As a homologous series is ascended, the size of the molecule increases
- This has an effect on the physical properties, such as boiling point and density
Functional Groups
- Functional groups determine the physical and chemical properties of molecules
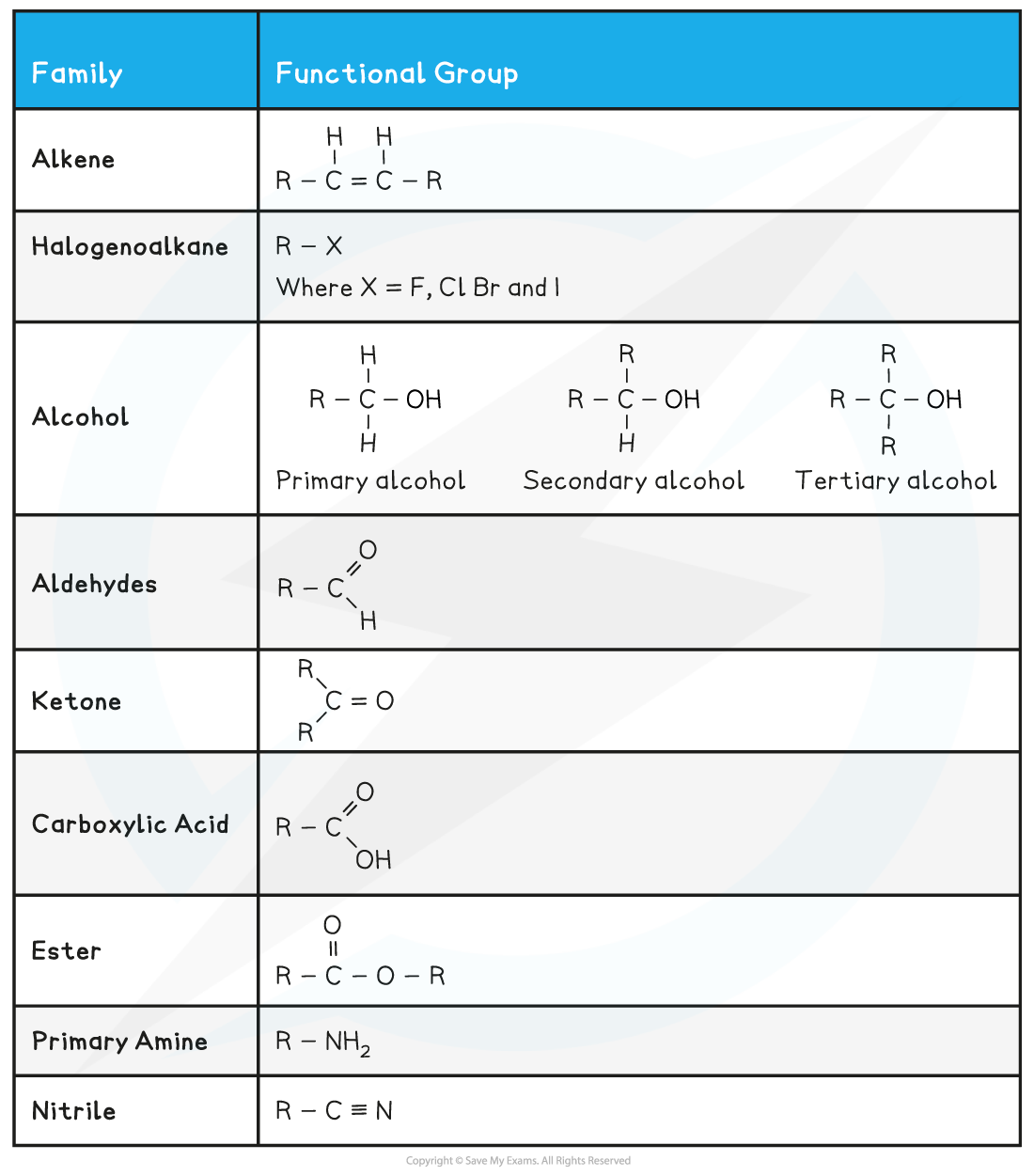
- The table below shows a summary of common functional groups found in compounds
- R is any other atom or group of atoms (except for hydrogen)
Functional Groups found in Compounds Table
Hydrocarbons
- Hydrocarbons are compounds that are made up of carbon and hydrogen atoms ONLY
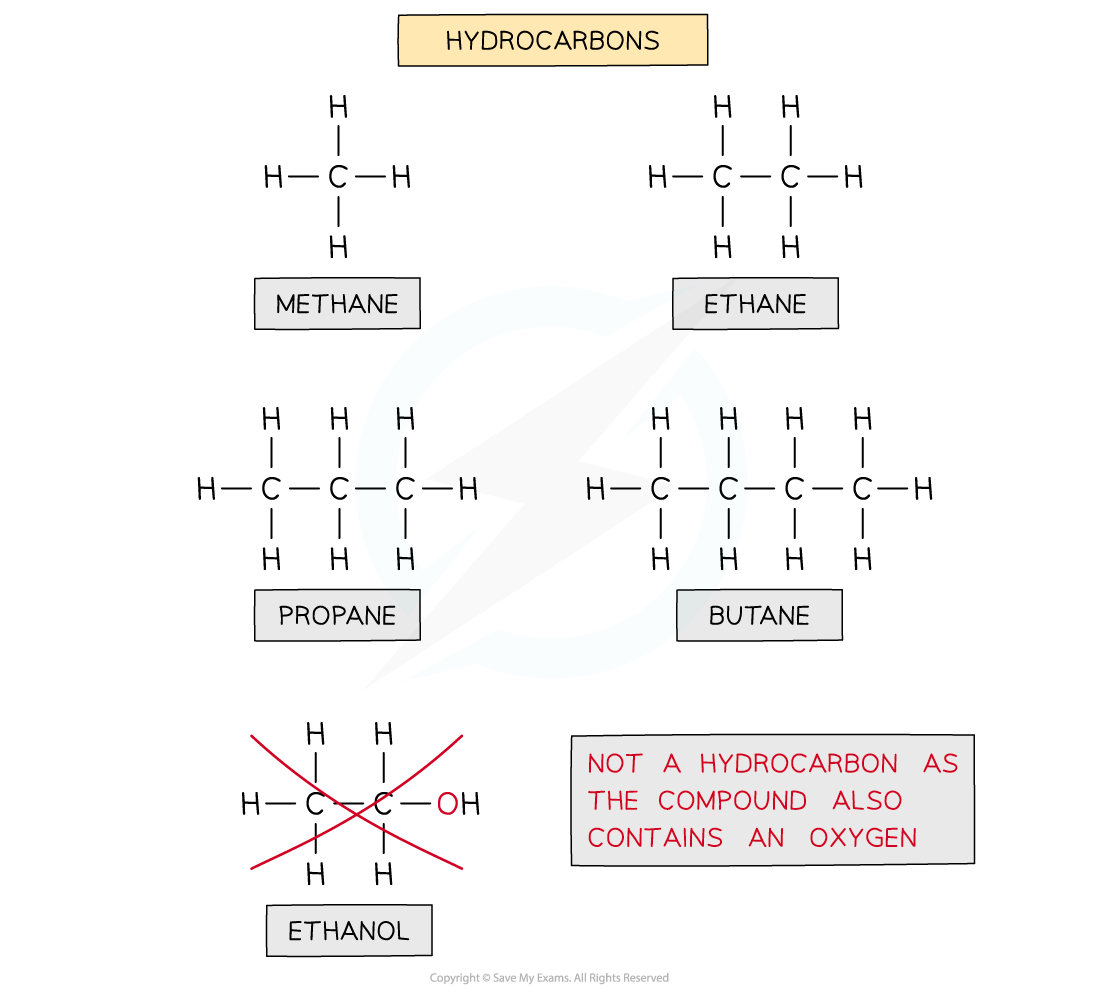
The diagram shows examples of hydrocarbons
Ethanol is NOT a hydrocarbon as the molecule also contains an oxygen atom and is not solely made up of carbon and hydrogen
转载自savemyexams
站内搜索
竞赛真题免费下载(点击下载)
在线登记
最新发布
© 2024. All Rights Reserved. 沪ICP备2023009024号-1





